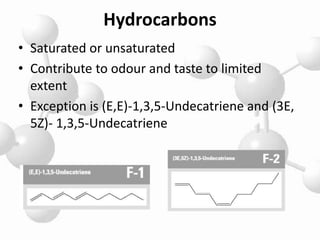
Aliphatic ingredients are important components in perfumery. They include hydrocarbons, alcohols, aldehydes, ketones, acids, and esters which are derived from straight carbon chains and contribute various odor characteristics. Key aliphatic ingredients used in perfumery include geraniol and citronellol which have floral notes, acetaldehyde and isobutyraldehyde which have fruity characteristics, and esters such as allyl amylglycolate which has a strong fruity galbanum odor and isoamyl butyrate which has a strongly fruity banana-like odor. Aliphatic ingredients provide many of the fundamental odor notes used in fragrance compositions.