1. Fine needle aspiration cytology (FNAC) of ovarian tumors can help differentiate between benign and malignant lesions and obtain a diagnosis. Specimens are prepared as direct smears, cytocentrifuge preparations, or cell blocks.
2. FNAC has limitations as the sensitivity is low between 26-40% and there is a high false negative rate, particularly for borderline tumors. However, it can confirm a clinical impression of a benign cyst.
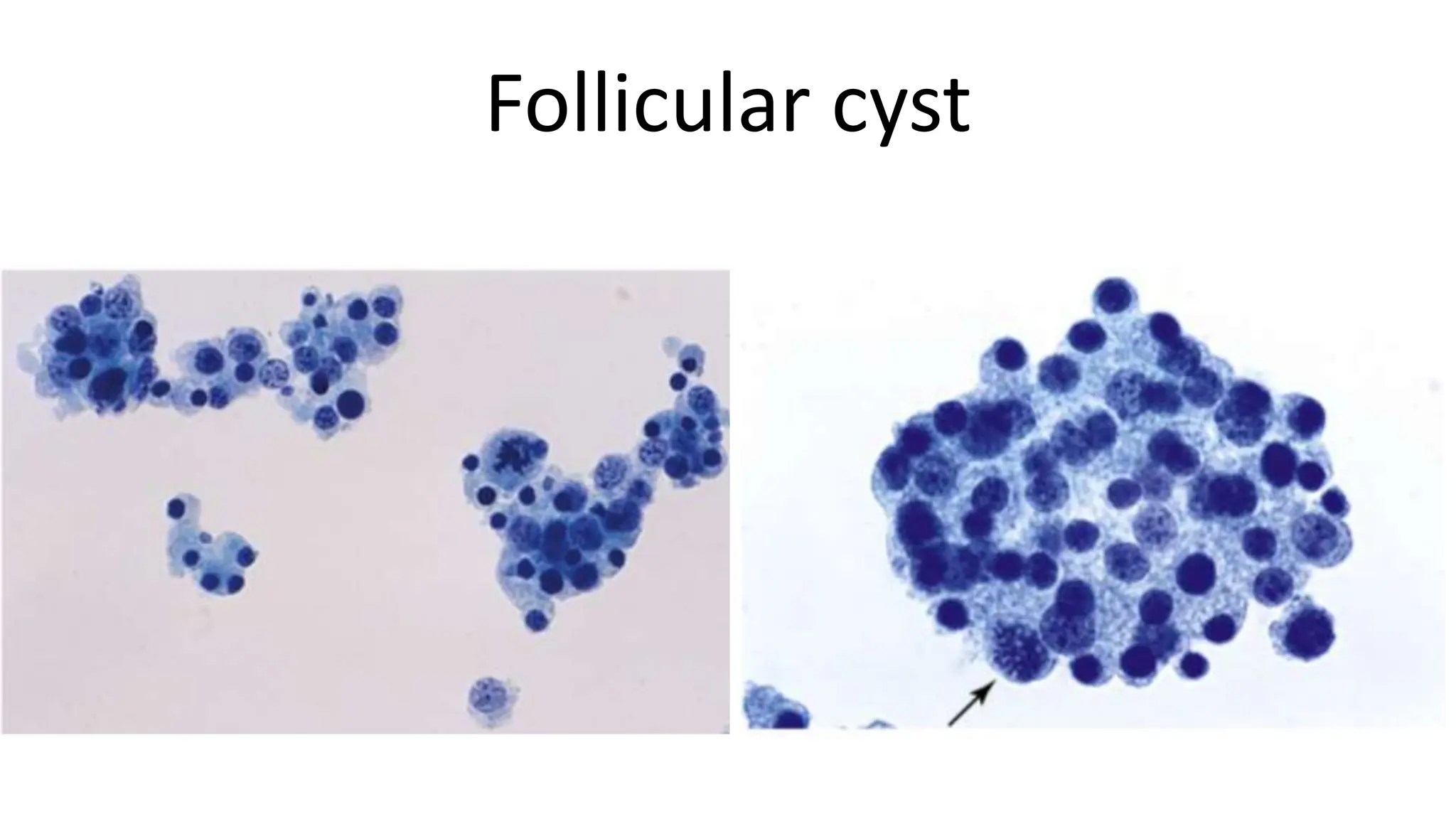
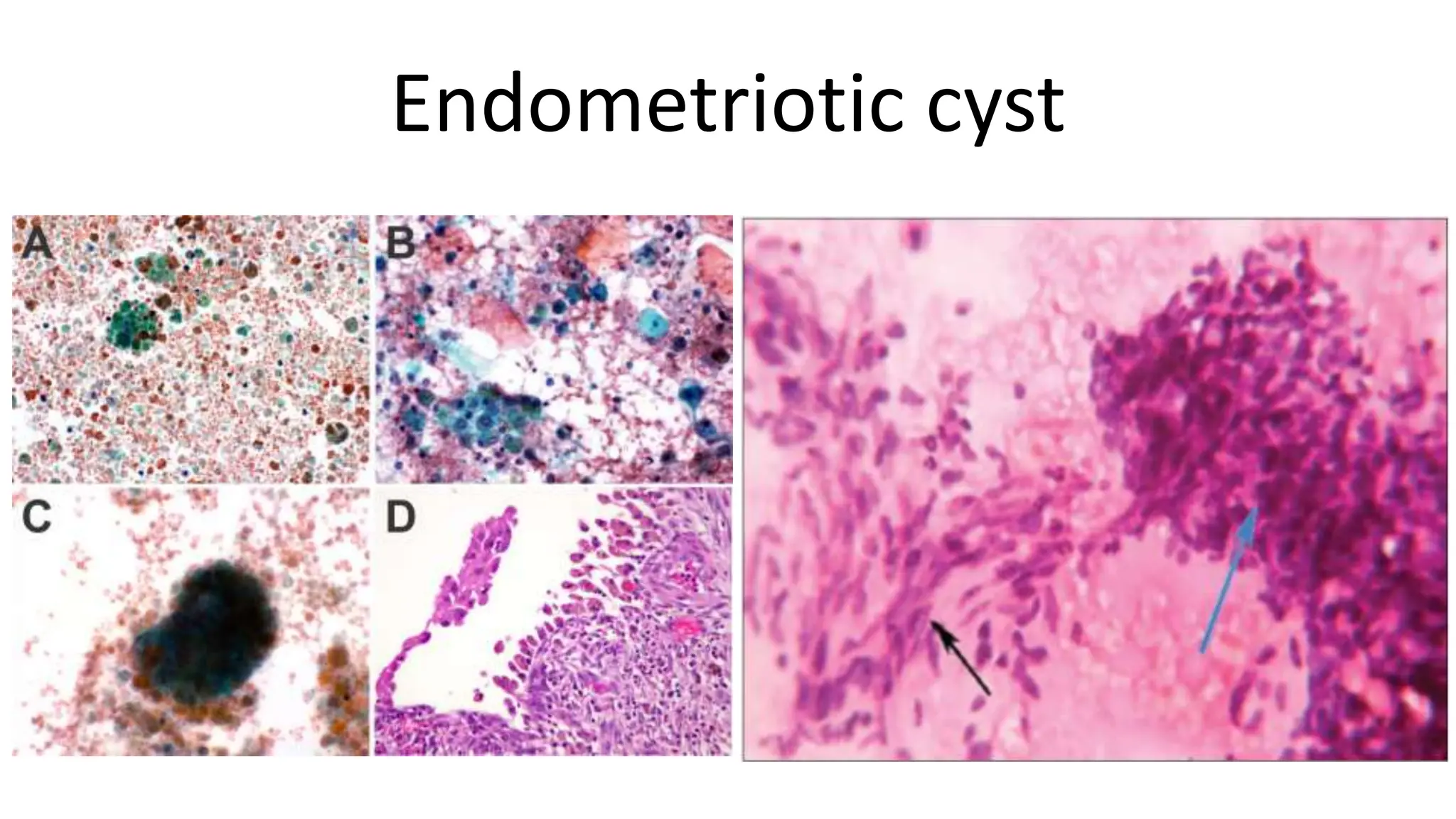
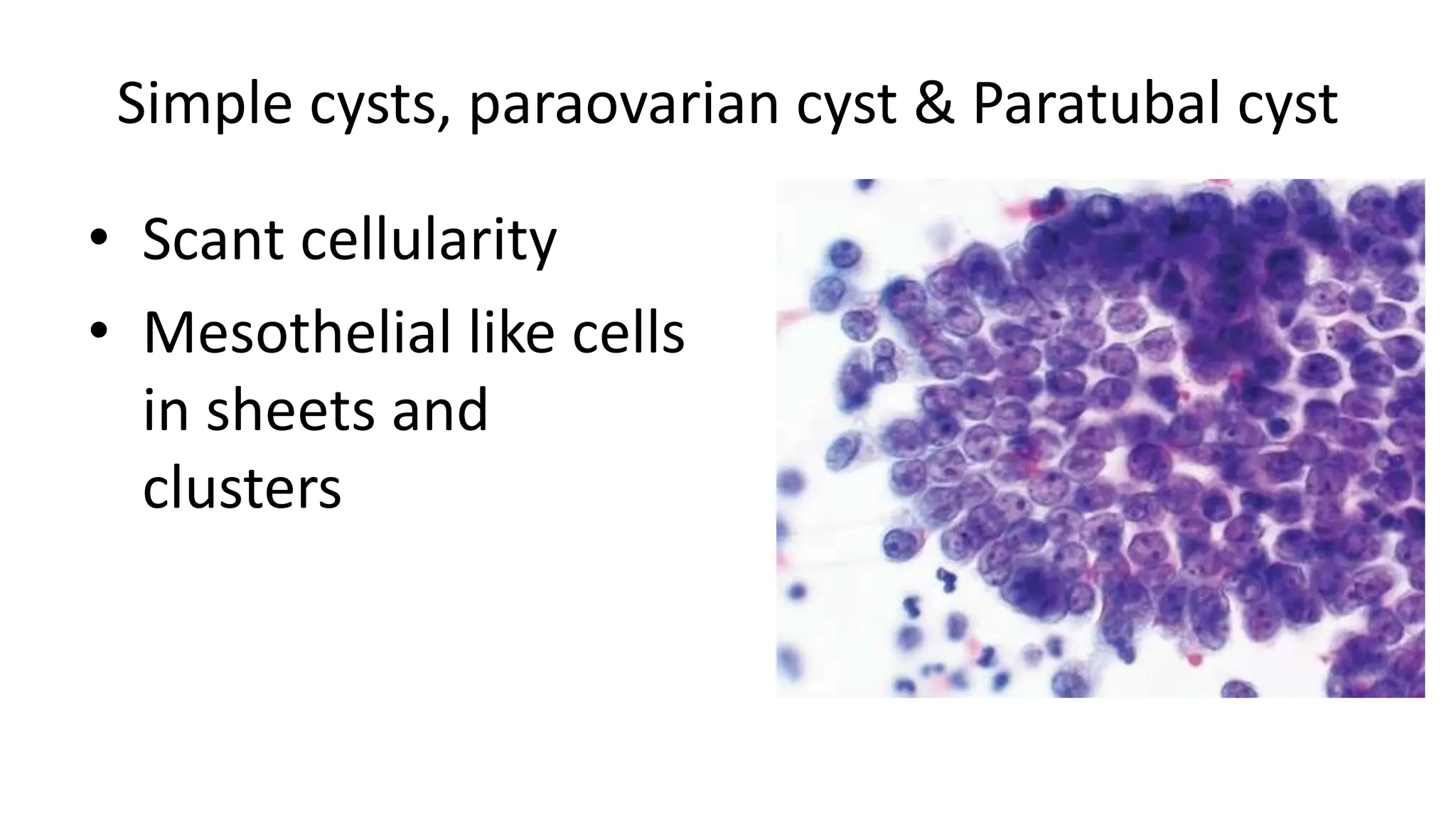
3. Different benign and malignant ovarian tumors have characteristic cytological features such as follicular cysts containing luteinized granulosa cells, serous cystadenomas containing cuboidal or ciliated cells, and serous carcinomas showing prominent nuclear



































![Yolk sac tumor
• [Resemble poorly differentiated carcinoma]
• Cellular smears
• Cohesive pleomorphic cells with prominent
nucleoli
• Intracytoplasmic and extracellular hyaline
globules](https://image.slidesharecdn.com/ahs-ovariantumorcytology-240127072311-644c31b8/75/AHS-Ovarian-tumor-Cytology-pptx-39-2048.jpg)















