Activity 2.nucleosynthesis
•Download as DOCX, PDF•
1 like•584 views
This document provides instructions for an activity to recall atomic symbols. Students are asked to complete a table distinguishing ions from isotopes. Ions are positively or negatively charged atoms that have the same number of protons but different numbers of electrons, while isotopes are atoms of the same element that have different atomic masses. Students are then asked questions to test their understanding, such as identifying which elements presented are ions based on their charges, which are isotopes based on having the same number of protons, and the differences between three isotopes of hydrogen.
Report
Share
Report
Share
Recommended
DESCRIBE THE GENERAL TYPES OF INTERMOLECULAR FORCES

A Lesson in Physical Science about Molecular Bonds for Grade 11 Students
Recommended
DESCRIBE THE GENERAL TYPES OF INTERMOLECULAR FORCES

A Lesson in Physical Science about Molecular Bonds for Grade 11 Students
Lesson 3- Synthesis of Elements in the Laboratory.pptx

Elements dont only discovered but also synthesize
Lesson 2 We Are All Made of Star Stuff (Formation of the Heavy Elements)

Content: How the elements found in the universe were formed
Content Standard:
At the end of the lesson, you will be able to demonstrate an understanding of:
the formation of the elements during the Big Bang and during stellar evolution
the distribution of the chemical elements and the isotopes in the universe
Learning Competencies:
At the end of the lesson,
Give evidence for and describe the formation of heavier elements during star formation and evolution (S11/12PS-IIIa-2)
Write the nuclear fusion reactions that take place in stars that lead to the formation of new elements (S11/12PS-IIIa-3)
Describe how elements heavier than iron are formed (S11/12PSIIIa-b-4))
Colors of light activity 1

A detailed lesson plan in Science 8
I. Objectives
At the end of the period, the student must be able to:
1. Perform the activity 1: Colors of the rainbow…colors of light
2. Identify the different colors of light after passing through the prism
3. Describe and give the reason behind the hierarchy of colors based on the observed results of the activity
4. Explain how refraction and dispersion takes place
Science 7 - Saturated and Unsaturated Solutions.pdf

PPT Presentation for Grade 7 Science - Saturated and Unsaturated Solutions.
More Related Content
What's hot
Lesson 3- Synthesis of Elements in the Laboratory.pptx

Elements dont only discovered but also synthesize
Lesson 2 We Are All Made of Star Stuff (Formation of the Heavy Elements)

Content: How the elements found in the universe were formed
Content Standard:
At the end of the lesson, you will be able to demonstrate an understanding of:
the formation of the elements during the Big Bang and during stellar evolution
the distribution of the chemical elements and the isotopes in the universe
Learning Competencies:
At the end of the lesson,
Give evidence for and describe the formation of heavier elements during star formation and evolution (S11/12PS-IIIa-2)
Write the nuclear fusion reactions that take place in stars that lead to the formation of new elements (S11/12PS-IIIa-3)
Describe how elements heavier than iron are formed (S11/12PSIIIa-b-4))
Colors of light activity 1

A detailed lesson plan in Science 8
I. Objectives
At the end of the period, the student must be able to:
1. Perform the activity 1: Colors of the rainbow…colors of light
2. Identify the different colors of light after passing through the prism
3. Describe and give the reason behind the hierarchy of colors based on the observed results of the activity
4. Explain how refraction and dispersion takes place
Science 7 - Saturated and Unsaturated Solutions.pdf

PPT Presentation for Grade 7 Science - Saturated and Unsaturated Solutions.
What's hot (20)
Lesson 3- Synthesis of Elements in the Laboratory.pptx

Lesson 3- Synthesis of Elements in the Laboratory.pptx
Lesson 2 We Are All Made of Star Stuff (Formation of the Heavy Elements)

Lesson 2 We Are All Made of Star Stuff (Formation of the Heavy Elements)
Module 1 Origin and Structure of the Earth Planet Earth.pptx

Module 1 Origin and Structure of the Earth Planet Earth.pptx
Science 7 - Saturated and Unsaturated Solutions.pdf

Science 7 - Saturated and Unsaturated Solutions.pdf
Distinction of Newton's First law and Galileo's Assertion

Distinction of Newton's First law and Galileo's Assertion
Similar to Activity 2.nucleosynthesis
Test bank for chemistry 4th edition by burdge

Test Bank for Chemistry 4th Edition by Burdge
Full download: https://goo.gl/bdpa6B
People also search:
business law: the first course - summarized case edition pdf
business law text and cases the first course pdf
business law the first course miller pdf
business law the first course summarized case edition 13th edition
miller business law text and cases the first course
business law the first course summarized case edition 14th edition
miller business law the first course summarized case edition
business law text and cases the first course 1st edition pdf
Wonders of Periodic Table

If all the elements are arranged in the order of their atomic weights, a periodic repetition of properties is obtained. This is expressed by the law of periodicity.— Dmitry Ivanovich Mendeleev
Wonders of periodic table

Periodic Table is the tabular arrangement of all the chemical elements which are organized based on atomic numbers, electronic configurations and existing chemical properties.
The Wonders of Periodic Table

If all the elements are arranged in the order of their atomic weights, a periodic repetition of properties is obtained. This is expressed by the law of periodicity.— Dmitry Ivanovich Mendeleev
Similar to Activity 2.nucleosynthesis (20)
Recently uploaded
A Strategic Approach: GenAI in Education

Artificial Intelligence (AI) technologies such as Generative AI, Image Generators and Large Language Models have had a dramatic impact on teaching, learning and assessment over the past 18 months. The most immediate threat AI posed was to Academic Integrity with Higher Education Institutes (HEIs) focusing their efforts on combating the use of GenAI in assessment. Guidelines were developed for staff and students, policies put in place too. Innovative educators have forged paths in the use of Generative AI for teaching, learning and assessments leading to pockets of transformation springing up across HEIs, often with little or no top-down guidance, support or direction.
This Gasta posits a strategic approach to integrating AI into HEIs to prepare staff, students and the curriculum for an evolving world and workplace. We will highlight the advantages of working with these technologies beyond the realm of teaching, learning and assessment by considering prompt engineering skills, industry impact, curriculum changes, and the need for staff upskilling. In contrast, not engaging strategically with Generative AI poses risks, including falling behind peers, missed opportunities and failing to ensure our graduates remain employable. The rapid evolution of AI technologies necessitates a proactive and strategic approach if we are to remain relevant.
How to Add Chatter in the odoo 17 ERP Module

In Odoo, the chatter is like a chat tool that helps you work together on records. You can leave notes and track things, making it easier to talk with your team and partners. Inside chatter, all communication history, activity, and changes will be displayed.
"Protectable subject matters, Protection in biotechnology, Protection of othe...

Protectable subject matters, Protection in biotechnology, Protection of other biological materials, Ownership and period of protection
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...National Information Standards Organization (NISO)
This presentation was provided by Steph Pollock of The American Psychological Association’s Journals Program, and Damita Snow, of The American Society of Civil Engineers (ASCE), for the initial session of NISO's 2024 Training Series "DEIA in the Scholarly Landscape." Session One: 'Setting Expectations: a DEIA Primer,' was held June 6, 2024.CACJapan - GROUP Presentation 1- Wk 4.pdf

Macroeconomics- Movie Location
This will be used as part of your Personal Professional Portfolio once graded.
Objective:
Prepare a presentation or a paper using research, basic comparative analysis, data organization and application of economic information. You will make an informed assessment of an economic climate outside of the United States to accomplish an entertainment industry objective.
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Let’s explore the intersection of technology and equity in the final session of our DEI series. Discover how AI tools, like ChatGPT, can be used to support and enhance your nonprofit's DEI initiatives. Participants will gain insights into practical AI applications and get tips for leveraging technology to advance their DEI goals.
Best Digital Marketing Institute In NOIDA

Safalta Digital marketing institute in Noida, provide complete applications that encompass a huge range of virtual advertising and marketing additives, which includes search engine optimization, virtual communication advertising, pay-per-click on marketing, content material advertising, internet analytics, and greater. These university courses are designed for students who possess a comprehensive understanding of virtual marketing strategies and attributes.Safalta Digital Marketing Institute in Noida is a first choice for young individuals or students who are looking to start their careers in the field of digital advertising. The institute gives specialized courses designed and certification.
for beginners, providing thorough training in areas such as SEO, digital communication marketing, and PPC training in Noida. After finishing the program, students receive the certifications recognised by top different universitie, setting a strong foundation for a successful career in digital marketing.
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.Digital Artifact 1 - 10VCD Environments Unit

Digital Artifact 1 - 10VCD Environments Unit - NGV Pavilion Concept Design
Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...

Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...Dr. Vinod Kumar Kanvaria
Exploiting Artificial Intelligence for Empowering Researchers and Faculty,
International FDP on Fundamentals of Research in Social Sciences
at Integral University, Lucknow, 06.06.2024
By Dr. Vinod Kumar Kanvariaclinical examination of hip joint (1).pdf

described clinical examination all orthopeadic conditions .
Normal Labour/ Stages of Labour/ Mechanism of Labour

Normal labor is also termed spontaneous labor, defined as the natural physiological process through which the fetus, placenta, and membranes are expelled from the uterus through the birth canal at term (37 to 42 weeks
Delivering Micro-Credentials in Technical and Vocational Education and Training

Explore how micro-credentials are transforming Technical and Vocational Education and Training (TVET) with this comprehensive slide deck. Discover what micro-credentials are, their importance in TVET, the advantages they offer, and the insights from industry experts. Additionally, learn about the top software applications available for creating and managing micro-credentials. This presentation also includes valuable resources and a discussion on the future of these specialised certifications.
For more detailed information on delivering micro-credentials in TVET, visit this https://tvettrainer.com/delivering-micro-credentials-in-tvet/
June 3, 2024 Anti-Semitism Letter Sent to MIT President Kornbluth and MIT Cor...

Letter from the Congress of the United States regarding Anti-Semitism sent June 3rd to MIT President Sally Kornbluth, MIT Corp Chair, Mark Gorenberg
Dear Dr. Kornbluth and Mr. Gorenberg,
The US House of Representatives is deeply concerned by ongoing and pervasive acts of antisemitic
harassment and intimidation at the Massachusetts Institute of Technology (MIT). Failing to act decisively to ensure a safe learning environment for all students would be a grave dereliction of your responsibilities as President of MIT and Chair of the MIT Corporation.
This Congress will not stand idly by and allow an environment hostile to Jewish students to persist. The House believes that your institution is in violation of Title VI of the Civil Rights Act, and the inability or
unwillingness to rectify this violation through action requires accountability.
Postsecondary education is a unique opportunity for students to learn and have their ideas and beliefs challenged. However, universities receiving hundreds of millions of federal funds annually have denied
students that opportunity and have been hijacked to become venues for the promotion of terrorism, antisemitic harassment and intimidation, unlawful encampments, and in some cases, assaults and riots.
The House of Representatives will not countenance the use of federal funds to indoctrinate students into hateful, antisemitic, anti-American supporters of terrorism. Investigations into campus antisemitism by the Committee on Education and the Workforce and the Committee on Ways and Means have been expanded into a Congress-wide probe across all relevant jurisdictions to address this national crisis. The undersigned Committees will conduct oversight into the use of federal funds at MIT and its learning environment under authorities granted to each Committee.
• The Committee on Education and the Workforce has been investigating your institution since December 7, 2023. The Committee has broad jurisdiction over postsecondary education, including its compliance with Title VI of the Civil Rights Act, campus safety concerns over disruptions to the learning environment, and the awarding of federal student aid under the Higher Education Act.
• The Committee on Oversight and Accountability is investigating the sources of funding and other support flowing to groups espousing pro-Hamas propaganda and engaged in antisemitic harassment and intimidation of students. The Committee on Oversight and Accountability is the principal oversight committee of the US House of Representatives and has broad authority to investigate “any matter” at “any time” under House Rule X.
• The Committee on Ways and Means has been investigating several universities since November 15, 2023, when the Committee held a hearing entitled From Ivory Towers to Dark Corners: Investigating the Nexus Between Antisemitism, Tax-Exempt Universities, and Terror Financing. The Committee followed the hearing with letters to those institutions on January 10, 202
Fresher’s Quiz 2023 at GMC Nizamabad.pptx

General quiz conducted for freshers ,2k23 batch at Government Medical College,Nizamabad
Contact:srisuryab199@gmail.com
How to Build a Module in Odoo 17 Using the Scaffold Method

Odoo provides an option for creating a module by using a single line command. By using this command the user can make a whole structure of a module. It is very easy for a beginner to make a module. There is no need to make each file manually. This slide will show how to create a module using the scaffold method.
Recently uploaded (20)
"Protectable subject matters, Protection in biotechnology, Protection of othe...

"Protectable subject matters, Protection in biotechnology, Protection of othe...
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...
Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion

Executive Directors Chat Leveraging AI for Diversity, Equity, and Inclusion
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...

Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...
Digital Artefact 1 - Tiny Home Environmental Design

Digital Artefact 1 - Tiny Home Environmental Design
Normal Labour/ Stages of Labour/ Mechanism of Labour

Normal Labour/ Stages of Labour/ Mechanism of Labour
Delivering Micro-Credentials in Technical and Vocational Education and Training

Delivering Micro-Credentials in Technical and Vocational Education and Training
June 3, 2024 Anti-Semitism Letter Sent to MIT President Kornbluth and MIT Cor...

June 3, 2024 Anti-Semitism Letter Sent to MIT President Kornbluth and MIT Cor...
How to Build a Module in Odoo 17 Using the Scaffold Method

How to Build a Module in Odoo 17 Using the Scaffold Method
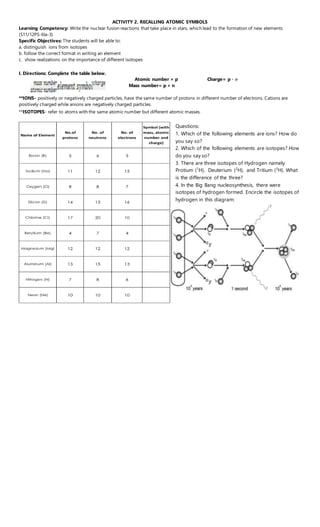
Activity 2.nucleosynthesis
- 1. ACTIVITY 2. RECALLING ATOMIC SYMBOLS Learning Competency: Write the nuclear fusion reactions that take place in stars, which lead to the formation of new elements (S11/12PS-IIIa-3) Specific Objectives: The students will be able to: a. distinguish ions from isotopes b. follow the correct format in writing an element c. show realizations on the importance of different isotopes I. Directions: Complete the table below. Atomic number = p Charge= p - e Mass number= p + n **IONS- positively or negatively charged particles, have the same number of protons in different number of electrons. Cations are positively charged while anions are negatively charged particles. **ISOTOPES- refer to atoms with the same atomic number but different atomic masses. Questions: 1. Which of the following elements are ions? How do you say so? 2. Which of the following elements are isotopes? How do you say so? 3. There are three isotopes of Hydrogen namely Protium (1 H), Deuterium (2 H), and Tritium (3 H). What is the difference of the three? 4. In the Big Bang nucleosynthesis, there were isotopes of hydrogen formed. Encircle the isotopes of hydrogen in this diagram:
