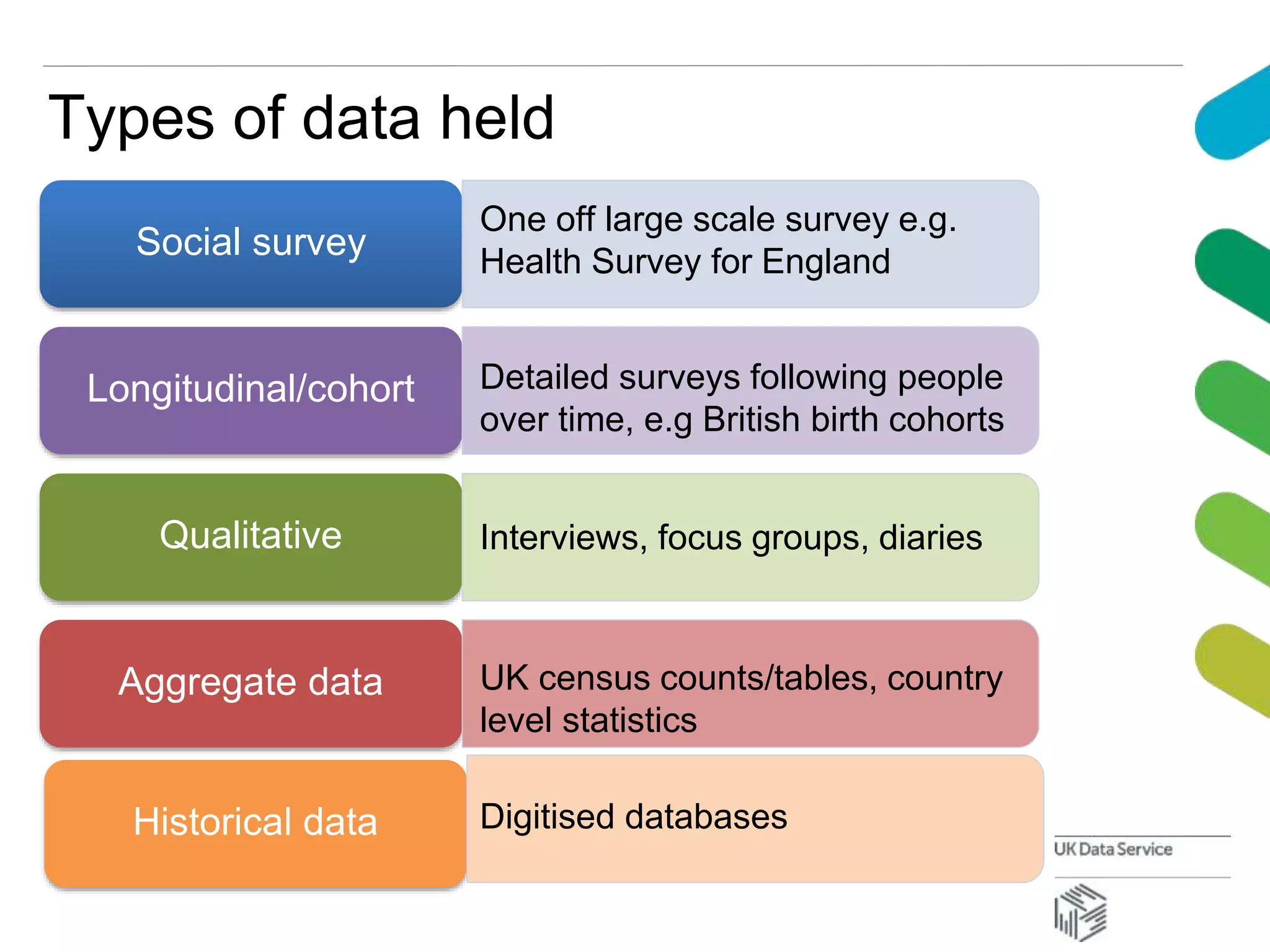
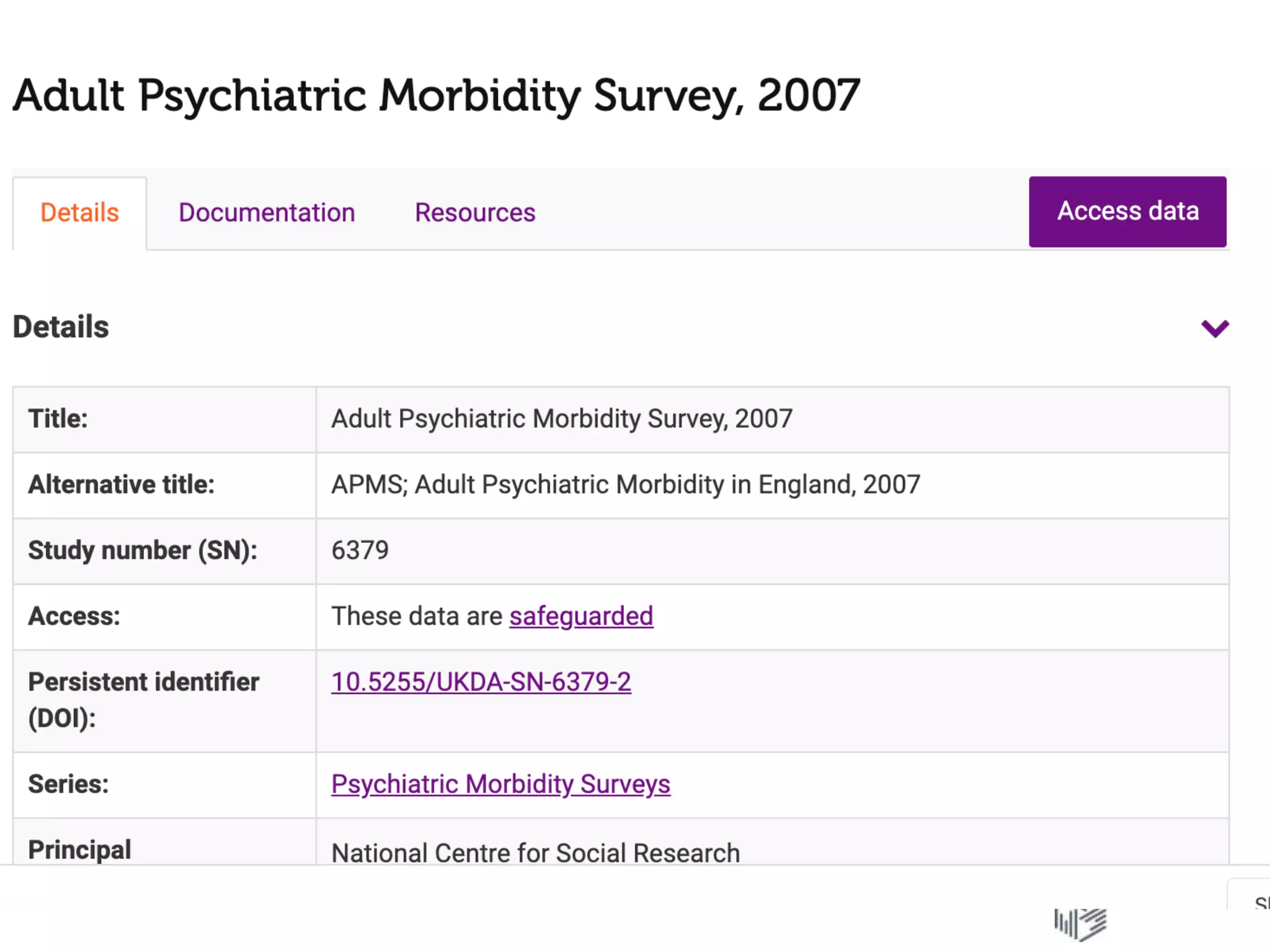
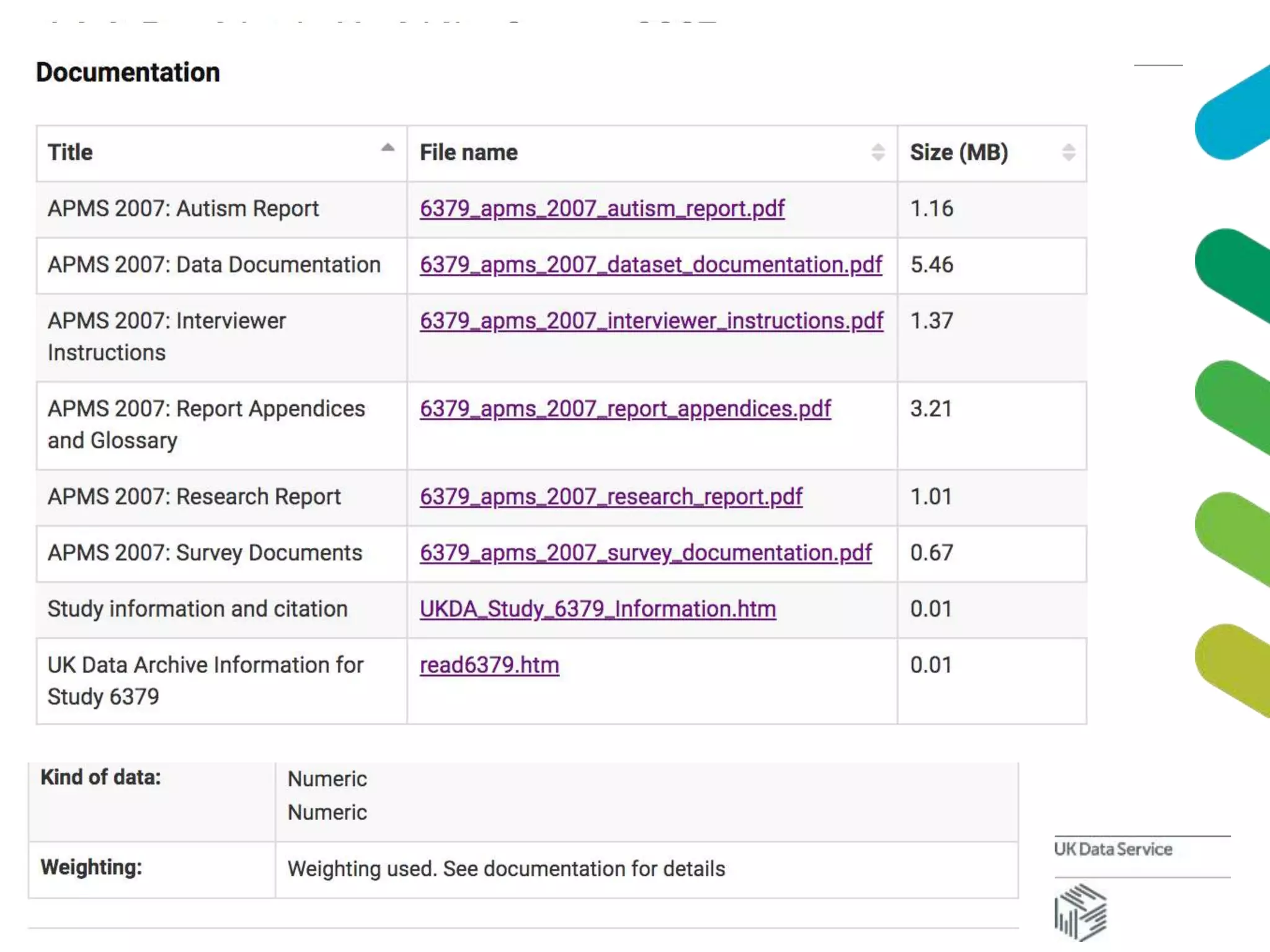
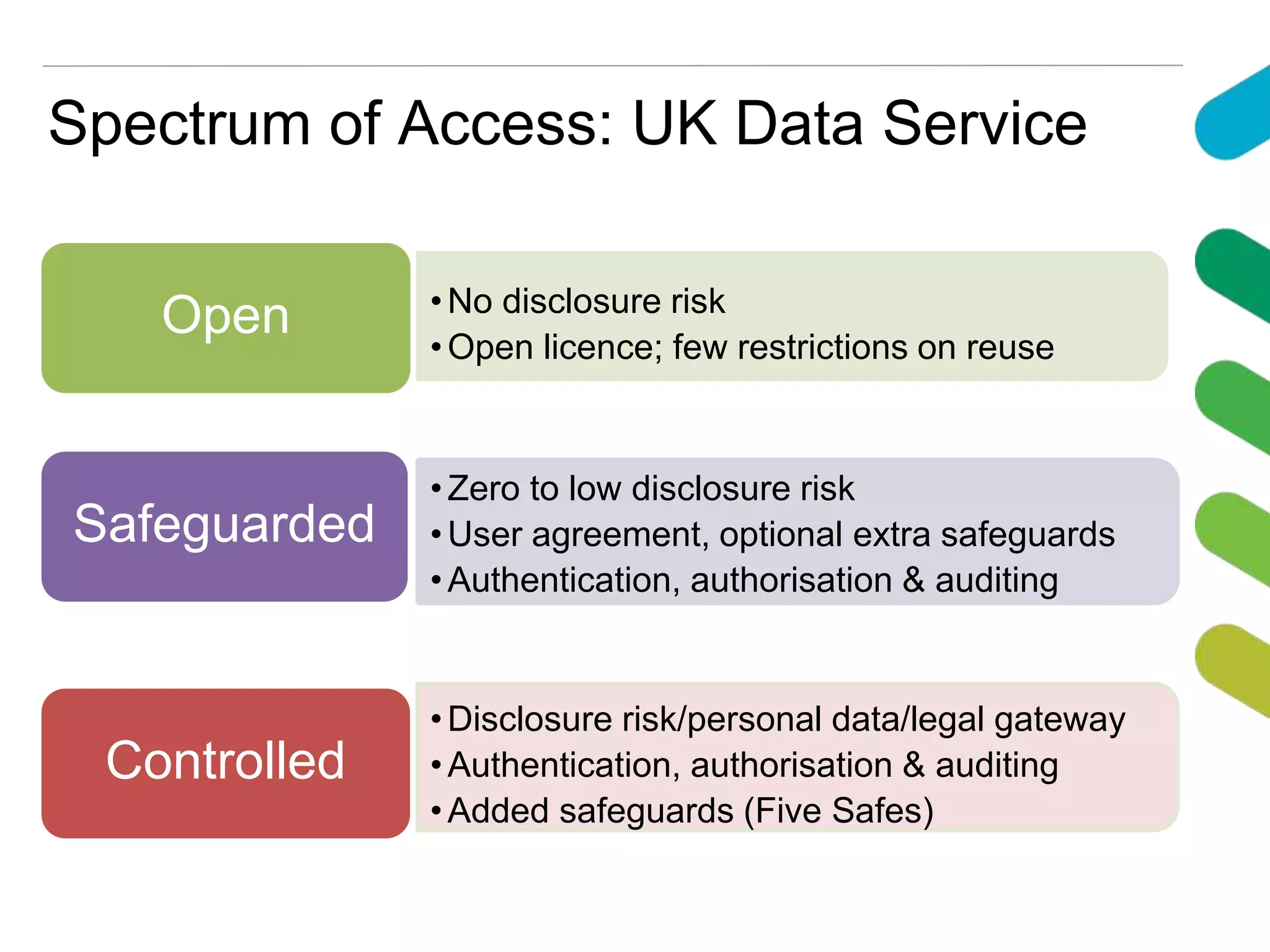
The document discusses data accessibility for research, focusing on the UK Data Service and its various data types, including health surveys and longitudinal studies. It outlines the processes for data publishing, management, and the safeguards in place to protect personal information through frameworks like the Five Safes. Additionally, it highlights the importance of compliance with data sharing policies and the challenges of identifying personal data within legal constraints.