1. The document provides information about systemic lupus erythematosus (SLE) including its epidemiology, symptoms, organ involvement, diagnostic tests, and disease management.
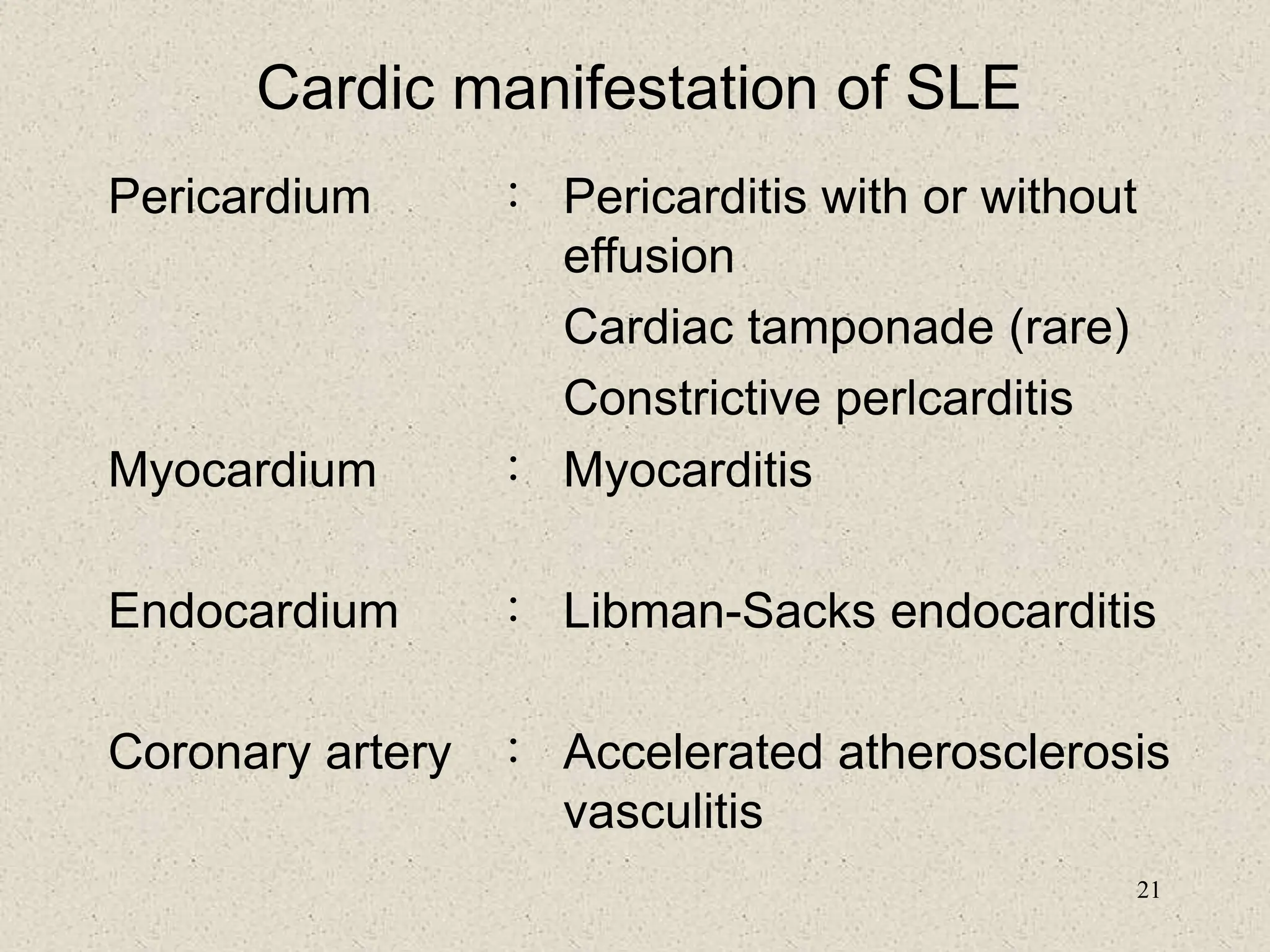
2. SLE most commonly affects women of childbearing age and has a variety of clinical manifestations involving the skin, joints, kidneys, and other organ systems.
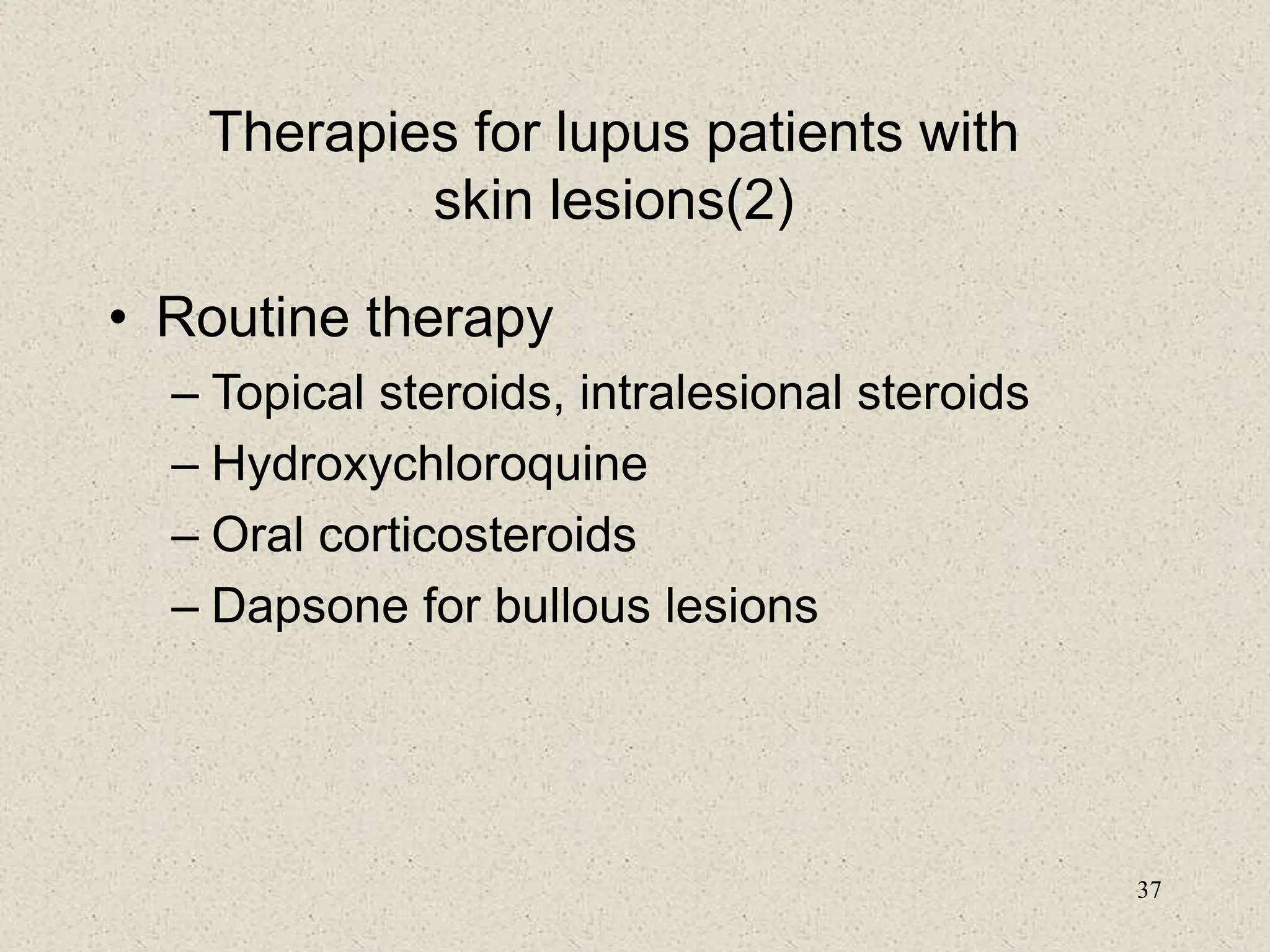
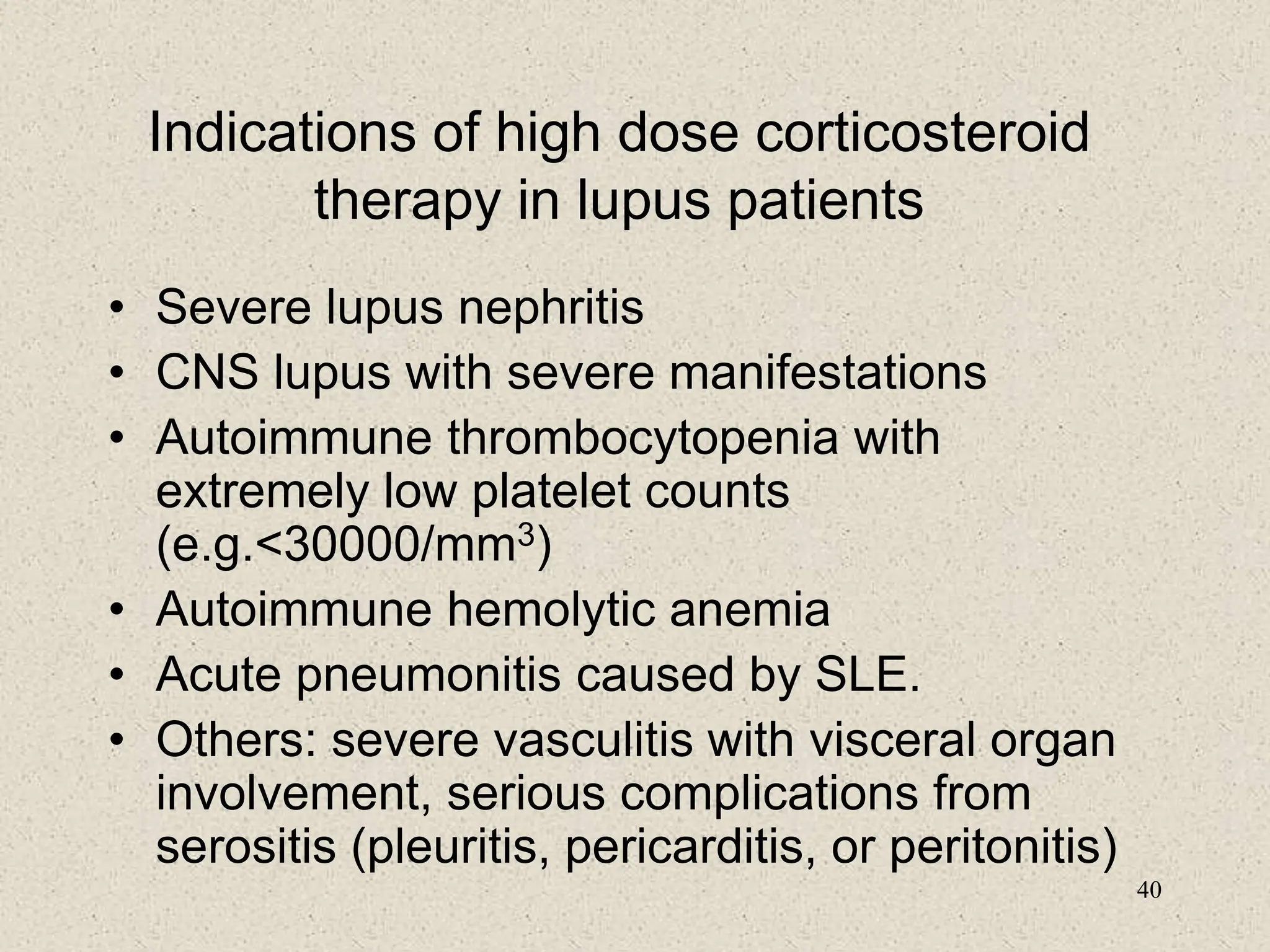
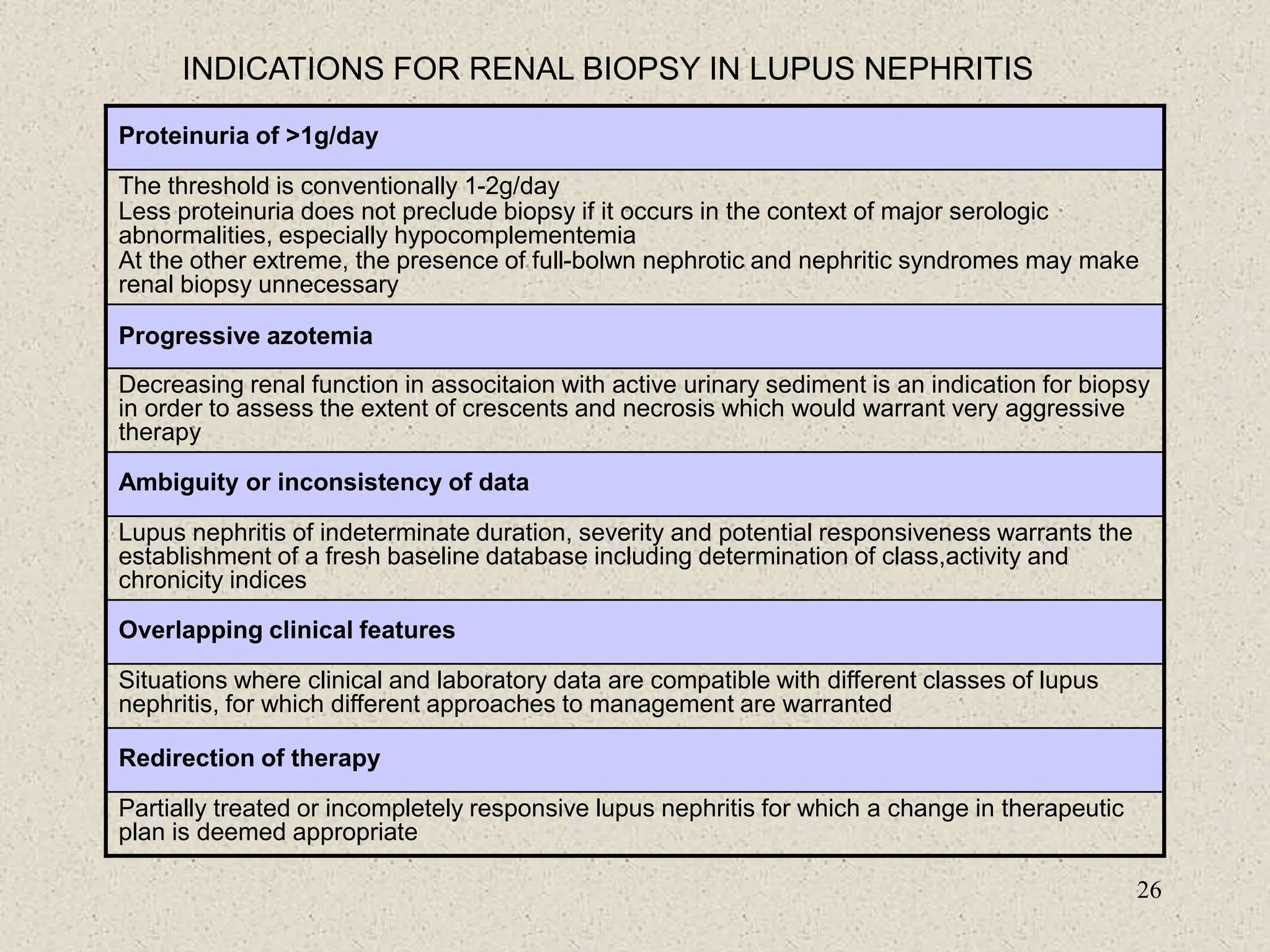
3. Diagnosis involves assessing clinical signs and symptoms along with serological tests like ANA and anti-DNA antibodies. Disease activity and organ involvement help guide treatment approaches.





























![32
FREQUENCY OF ABNORMAL LABORATORY TESTS COMMONLY USED IN THE
EVALUATION OF NEUROPSYCHIATRIC LUPUS ERYTHEMATOSUS
Test Frequency of
Abnormal Test Result
Range (%)
Comment
Serologic
Antimeuronal antibodies
Antineurofilament antibodies
Antiribosomal-P antibodies
Antiphospholipid antibodies
30-92
58
45-90
45-80
Diffuse manifestations
Diffuse manifestations
Psychosis/depression
Focal manifestations, strokes
Cerebrospinal fluid
Routine
Pleocytosis
Increased protein]
Low glucose
6-34
22-50
3-8
Rule out infection and NSAID meningitis
Nonspecific
Rule out infection, transerse myelitis
Special
Antineuronal antibodies (lgG)
Elevated Q albumin
Elevated lgG/lgM index
Oligoclonal bands (2 bands)
90
8-33
25-66
20-82
Diffuse manifestations, present in 40%
with focal manifestations
Break in blood-brain barrier
Diffuse manifestations
Diffuse manifestations](https://image.slidesharecdn.com/8-guidelineforslemanagement-240116024039-4ace0a34/75/8-Guideline-for-elaborate-SLE-management-ppt-32-2048.jpg)