4 volumetric analysis learning outcomes
•Download as DOCX, PDF•
2 likes•6,119 views
Report
Share
Report
Share
Recommended
Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...

Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...Dr. Pradeep mitharwal
OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...

OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...Ratnakaram Venkata Nadh
More Related Content
What's hot
Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...

Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...Dr. Pradeep mitharwal
OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...

OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...Ratnakaram Venkata Nadh
What's hot (20)
Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...

Synthesis, Characterization, Spectral (FT-IR, 1H, 13C NMR, Mass and UV) and B...
Determination of enthalpy of ionisation of acetic acid

Determination of enthalpy of ionisation of acetic acid
Mechanistic Aspects of Oxidation of P-Bromoacetophen one by Hexacyanoferrate ...

Mechanistic Aspects of Oxidation of P-Bromoacetophen one by Hexacyanoferrate ...
OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...

OXIDATION OF POLYETHYLENE GLYCOL-200 BY POTASSIUM PERIODATE IN ALKALINE MEDIU...
Mechanism of the Reaction of Plasma Albumin with Formaldehyde in Ethanol - Wa...

Mechanism of the Reaction of Plasma Albumin with Formaldehyde in Ethanol - Wa...
Synthesis and characterization of new derivatives of thiazole with liquid cry...

Synthesis and characterization of new derivatives of thiazole with liquid cry...
ORGANIC CHEMISTRY QUESTIONS FOR ADVANCED LEVEL STUDENTS

ORGANIC CHEMISTRY QUESTIONS FOR ADVANCED LEVEL STUDENTS
Viewers also liked
Viewers also liked (20)
Similar to 4 volumetric analysis learning outcomes
Similar to 4 volumetric analysis learning outcomes (20)
PPT Preparation of Solutions and Bench Reagents.pdf

PPT Preparation of Solutions and Bench Reagents.pdf
More from Martin Brown
More from Martin Brown (20)
Jcsp library project Reading promotion - 10 great opening lines

Jcsp library project Reading promotion - 10 great opening lines
Jcsp library project Reading promotion - 10 great halloween reads - poster

Jcsp library project Reading promotion - 10 great halloween reads - poster
Recently uploaded
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Recently uploaded (20)
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
4 volumetric analysis learning outcomes
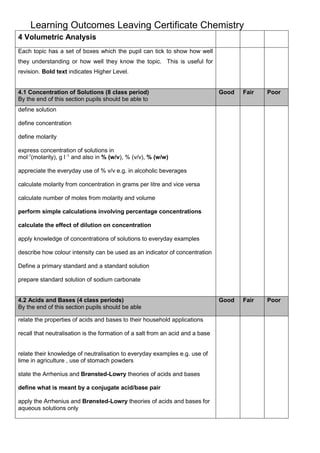
- 1. Learning Outcomes Leaving Certificate Chemistry 4 Volumetric Analysis Each topic has a set of boxes which the pupil can tick to show how well they understanding or how well they know the topic. This is useful for revision. Bold text indicates Higher Level. 4.1 Concentration of Solutions (8 class period) By the end of this section pupils should be able to Good Fair Poor Good Fair Poor define solution define concentration define molarity express concentration of solutions in mol-1(molarity), g l -1 and also in % (w/v), % (v/v), % (w/w) appreciate the everyday use of % v/v e.g. in alcoholic beverages calculate molarity from concentration in grams per litre and vice versa calculate number of moles from molarity and volume perform simple calculations involving percentage concentrations calculate the effect of dilution on concentration apply knowledge of concentrations of solutions to everyday examples describe how colour intensity can be used as an indicator of concentration Define a primary standard and a standard solution prepare standard solution of sodium carbonate 4.2 Acids and Bases (4 class periods) By the end of this section pupils should be able relate the properties of acids and bases to their household applications recall that neutralisation is the formation of a salt from an acid and a base relate their knowledge of neutralisation to everyday examples e.g. use of lime in agriculture , use of stomach powders state the Arrhenius and Brønsted-Lowry theories of acids and bases define what is meant by a conjugate acid/base pair apply the Arrhenius and Brønsted-Lowry theories of acids and bases for aqueous solutions only
- 2. Learning Outcomes Leaving Certificate Chemistry 4.3 Volumetric Analysis (22 class Periods) By the end of this section pupils should be able identify appropriate apparatus used in volumetric analysis use correct titrimetric procedure when carrying out titrations solve volumetric problems, using the formula method solve volumetric problems from first principles carry out a titration between hydrochloric acid and sodium hydroxide solutions and use this titration to make a sample of sodium chloride (OL only) standardise a hydrochloric acid solution using a standard solution of sodium carbonate calculate the relative molecular mass of a compound and of the amount of water of crystallisation in a compound from titration data (balanced equations will be given in all volumetric problems) determine the concentration of ethanoic acid in vinegar determine the amount of water of crystallisation in hydrated sodium carbonate carry out a potassium manganate(VII)/ammonium iron(II) sulfate titration determine the amount of iron in an iron tablet carry out an iodine/thiosulfate titration determine the percentage (w/v) of hypochlorite in bleach Good Fair Poor