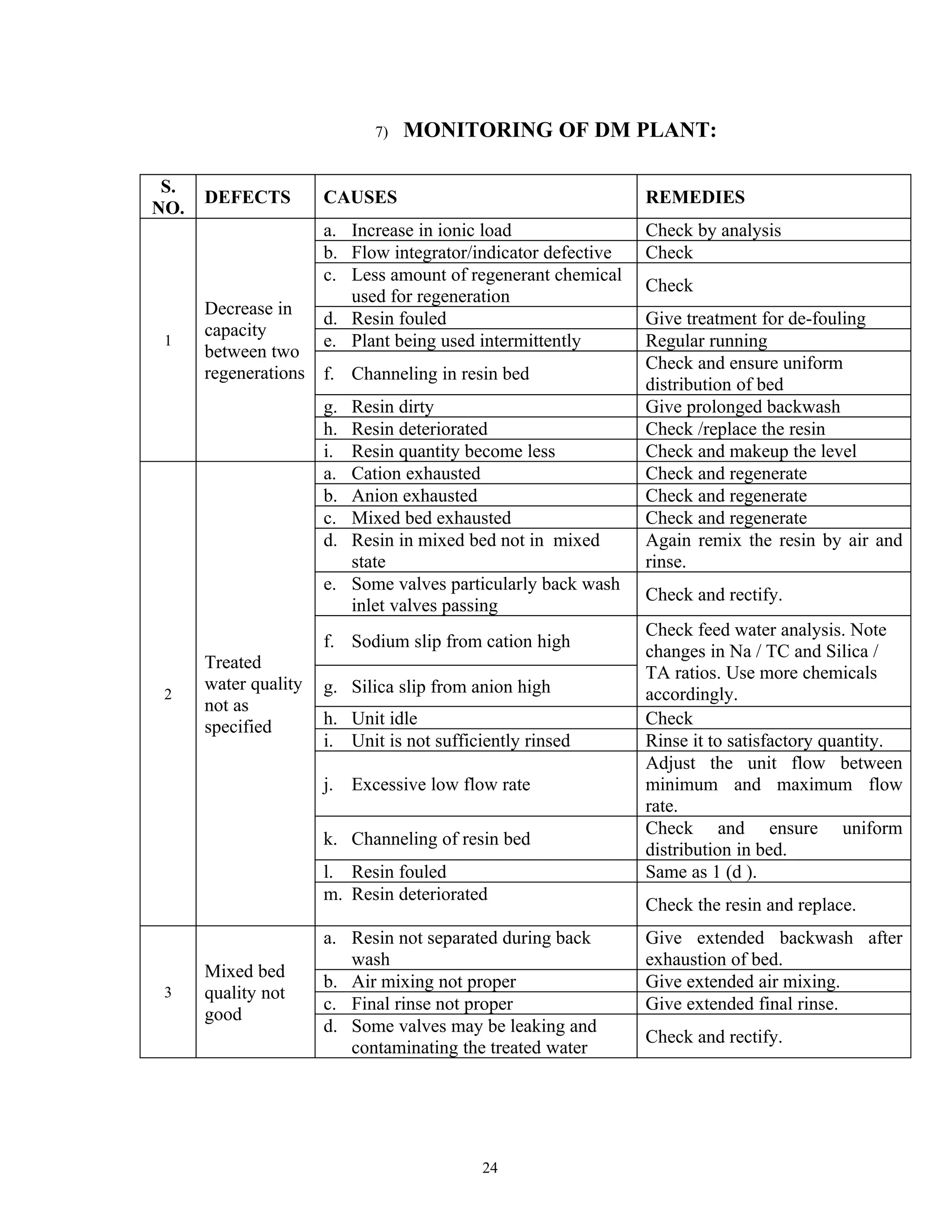
The document discusses raw water impurities and demineralization systems. It begins by describing common impurities found in raw water such as calcium, magnesium, bicarbonates, sulfates and chlorides. It then discusses methods for expressing dissolved impurities and difficulties caused by various impurities. The document outlines pretreatment processes including coagulation, settling and filtration to remove suspended solids. It also describes demineralization systems using ion exchange materials like cation and anion exchange resins. Different demineralization configurations are listed that can be used depending on raw water quality and purity requirements.








![2) METHOD OF EXPRESSING DISSOLVED IMPURITIES:
Dissolved impurities may be expressed in terms of ions themselves or in terms of
their equivalent. The preferred method of expression in water treatment field is in
terms of equivalent of Calcium Carbonate abbreviated ‘as CaCo3.’
CaCo3 is a good common denominator because it has a molecular weight of 100,
which facilitates calculations. Moreover in this form of analysis the sum of cations
always equal to sum of anions. This method also aids in predicting the comparative
analysis after various forms of treatment and analysis at consecutive steps in multi
step demineralization.
If analysis expressed in terms of ions, it can be converted to the form of expression in
terms of equivalent CaCo3 (or as CaCo3 ) by dividing figures by equivalent weight of
ions then multiplying by equivalent weight of CaCo3 (i.e. 50).
For example if amount of Calcium in water is 40 ppm as Ca then during expressing it
in terms of CaCo3 it becomes
40
----- X 50 = 100 ppm as Caco3. (Here 20 is equivalent wt. of Ca)
20
(Equivalent weight of an ion is its molecular weight divided by its valances)
Other units of analysis of water are:
a) one grain/u.s. gallon = 17.1 ppm
b) one grain/imperial gallon = 14.3 ppm
c) one milligram/liter = 1 ppm
Or
one gram/m3 = 1 ppm
[Note 1 U.S. gallon = 8.33 pounds
1 pound = 7000 grams
1 imperial gallon = 10 pounds
1 liter = 1,000,000 mg]
9](https://image.slidesharecdn.com/23291582-dm-plant-120623095455-phpapp02/75/23291582-dm-plant-9-2048.jpg)


















![A maximum endpoint value of 0.3 mg/1 SiO2 above the average leakage should not be
exceeded in order to avoid a high contamination of the polishing resin layer and unacceptably
high silica leakage during subsequent cycles. Silica leakage can be minimized by operating
the plant at silica break rather than conductivity end point. This secures the lowest silica
leakage, but at the expense of a 5 – 10 % throughput reduction.
8.2 SELECTION OF LAYOUT & RESIN TYPES:
The plant configuration will depend on the feed water composition, the water quality
required and the economics of operation. The following general guidelines are given to help
in configuration and resin selection.
(A) [SAC] – [WBA]: This combination of strong acid cation [SAC] and weak base
anion [WBA] resins is used to obtain partially deionized water without removal of
CO2 and SiO2.
(B) [SAC] – [SBA]: The combination of strong acid cation and strong base anion [SBA]
is preferred for treating low mineralized water or for small size plants.
(C) [SAC] – [WBA] – [SBA] : This combination of strong acid cation with weak base
and strong base anions is proven to be an excellent choice for larger plants as it
provides an optimum balance between investment and running cost. It is well suited
to treat waters with low alkalinity, when the FMA (Cl + NO3 + SO4) is typically >
60% of the total anions. The normal end-point for a WBA resin corresponds to the
chloride breakthrough, which means that the downstream SBA resin is only
removing the carbon dioxide and silica ions. This situation generally leads to a big
discrepancy between WBA (large) and SBA (low) volumes.
(D) [WAC] – [SAC] – [SBA]: The use of a weak acid cation [WAC] in front of a strong
anion is preferred with feed waters containing a high proportion of temporary
hardness (>60%) and low FMA. The normal end-point for a WAC resin is 10%
alkalinity leak. In this condition, the down-stream SAC resin should remove the
permanent hardness and the monovalent cations. This situation generally leads to a
big discrepancy between WAC (large). This is the ideal combination for high
hardness, high alkalinity and high FMA water, as well as large size plants. Again
the cation and anion combinations can be in single or separate vessels.
29](https://image.slidesharecdn.com/23291582-dm-plant-120623095455-phpapp02/75/23291582-dm-plant-29-2048.jpg)
![(E) [WAC] – [SAC] – [WBA] – [SBA]: This is the ideal combination for high hardness,
high alkalinity & high FMA water, as well as large size plants. Again the Cation &
Anion combination can be single or separate vessel.
8.3 ATMOSPHERIC DEGASIFIER:
The decision to install an atmospheric degasifier is based principally on economical
considerations. Removing carbon dioxide before it reaches the anion resins will reduce
NaOH chemical consumption stoichiometrically and this should be balanced against the cost
of the degasifier. Generally the economical balance is not in favor of a degasifier for small
plants (up to about 10 m3/h or 50 gpm). For larger plants, if the total CO2 is greater than 80-
100 mg/1 (ppm), the pay-back time for a degasifier should be short. For very large plants,
the limit can be reduced to 50 mg/l CO2.
8.4 OUTPUT BASED ON WATER QUALITY:
Output of DM plant is depending on water quality, if water quality vary output will also
changed accordingly. So regular monitoring of raw water quality is required and accordingly
output may be calculated.
Some time conductivity of anion remains high since initial stage of service run. This may due
to either CaSO4 precipitation on SAC, Organic fouling & silica deposit on anion resin.
30](https://image.slidesharecdn.com/23291582-dm-plant-120623095455-phpapp02/75/23291582-dm-plant-30-2048.jpg)