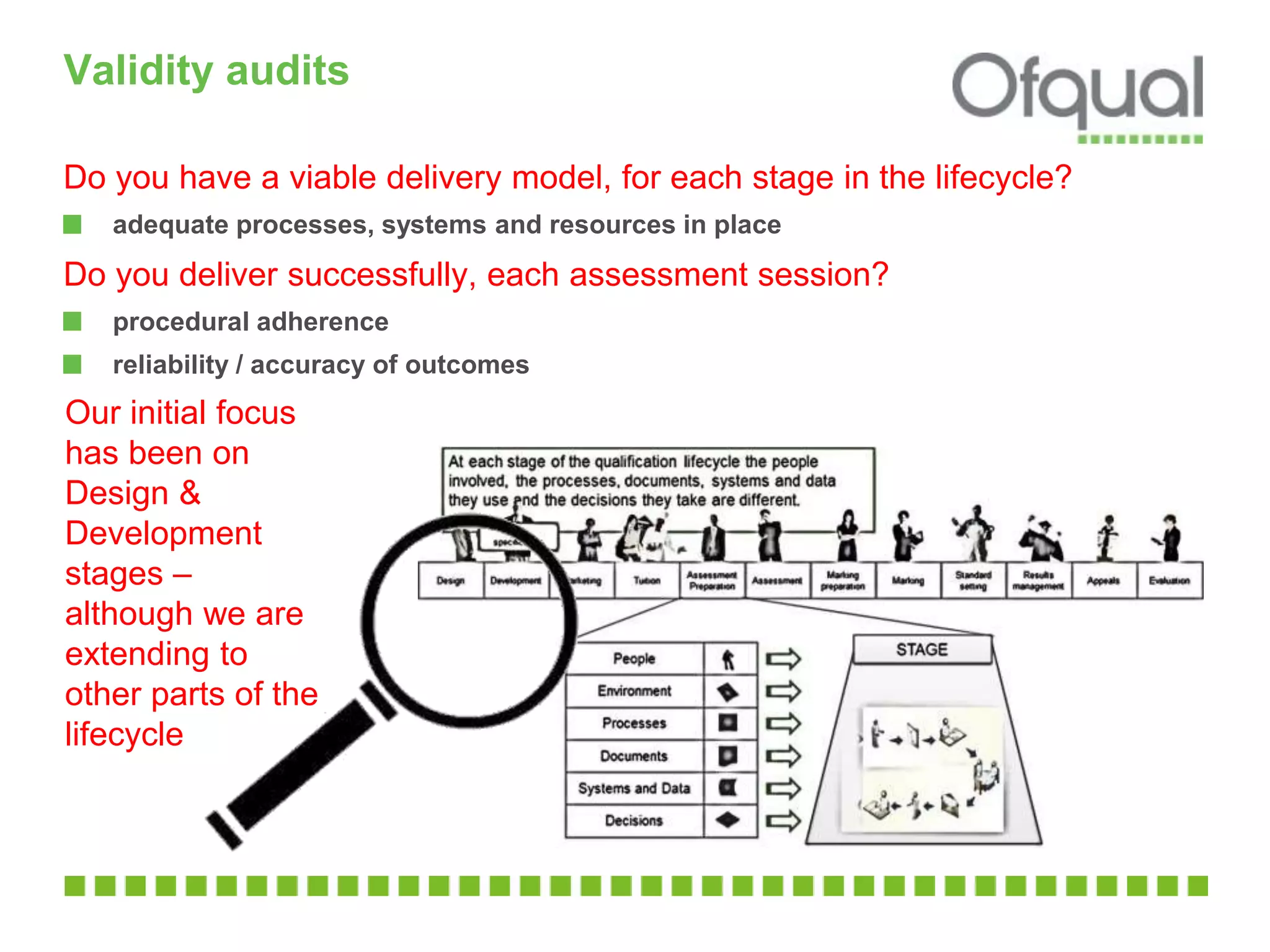
The document discusses validity audits that will be conducted by an organization on awarding organizations. The audits will be scheduled both randomly and based on risk factors like the organization, qualification type, and stage in the qualification lifecycle. Priorities will be publicly funded qualifications, those taken by many students, and those used in apprenticeships. Audits will review documentation and processes around designing, developing, and delivering qualifications to ensure they are valid and fit for purpose. Key areas that will be examined include the objective and support for the qualification, defining required skills, developing assessments, setting and maintaining standards, and monitoring achievement of objectives. Organizations should ensure all current qualifications are valid and be prepared to demonstrate their approach to setting and maintaining