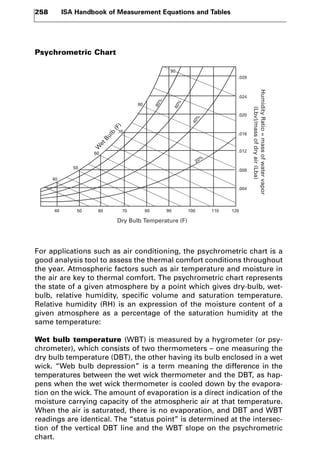
This document discusses principles and methods of humidity measurement. It defines key terms like relative humidity, wet bulb temperature, and dew point. Popular devices for measurement include hygrometers, thermohygrometers, psychrometers, and dew point meters. Thermohygrometers typically use a sponge or electronic sensor to measure humidity along with a thermometer to measure temperature. Psychrometers use two thermometers - a dry bulb and wet bulb, with the latter wrapped in a moist wick, to determine humidity through evaporation rates. Psychrometric charts graphically represent atmospheric conditions and are useful for applications like HVAC.



![Dalton’s Law of Partial Pressures
John Dalton’s law:
The Total Pressure of a gas mixture is the sum of the pressures of each
gas component.
P = Pn2
+ Po2
+ Par + . . .
“Partial Pressure” is defined as the pressure of a single gas in the mix-ture
as if that gas alone occupied the container.
Water in its gaseous state (vapor) is an additional gas component of air,
and also appears in Dalton’s law as:
P = Pn2
+ PO2
+ PAr + PCO2
…… + e = Pda + e
where
e = partial pressure of (water) vapor [mbar]
Pda = partial pressure of dry air
Chapter 9/Humidity 255
Main Gas Components in Air
Gas % Volume % Weight
Nitrogen N2 78.03 75.47
Oxygen O2 20.99 23.20
Argon Ar 0.93 1.28
Carbon Dioxide CO2 0.03 0.04
All others: H2, He, Ne, Kr etc. 0.02 0.01](https://image.slidesharecdn.com/182409-140906080756-phpapp01/85/Instrumentation-and-control-4-320.jpg)