Cytolon's Patients as Partners Appraoch
•
0 likes•541 views
The document discusses a new model where patients are partners in clinical trials. It suggests that patients should be able to communicate with peers, investigators, and primary care physicians before, during, and after trials. It also proposes that patients own their individual trial data and that data can be donated or shared in a de-identified manner with researchers while still maintaining patient privacy.
Report
Share
Report
Share
Recommended
Setting Normative Function: Identification of areas where Standards and Guide...

Setting Normative Function: Identification of areas where Standards and Guide...Human Variome Project
Recommended
Setting Normative Function: Identification of areas where Standards and Guide...

Setting Normative Function: Identification of areas where Standards and Guide...Human Variome Project
Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"

Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
More Related Content
What's hot
Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"

Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"The Petrie-Flom Center for Health Law Policy, Biotechnology, and Bioethics
What's hot (13)
Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"

Holly Fernandez Lynch, "Career Opportunities in Health Law and Bioethics"
Whitepaper #5 the_data_mineinc_differentiate_or_dissipate

Whitepaper #5 the_data_mineinc_differentiate_or_dissipate
Women Wading Through the Web: providing women with the tools to evaluate onli...

Women Wading Through the Web: providing women with the tools to evaluate onli...
Developing Evidence-Based Guidelines for Patient-Accessible Medical Records

Developing Evidence-Based Guidelines for Patient-Accessible Medical Records
Viewers also liked
Viewers also liked (16)
CDAA Who Am I Meant to Be and Who have I Become__MBoult_10April2015

CDAA Who Am I Meant to Be and Who have I Become__MBoult_10April2015
Estudio de Necesidades y Estrategias de Endomarketing para una Microfinanciera

Estudio de Necesidades y Estrategias de Endomarketing para una Microfinanciera
Similar to Cytolon's Patients as Partners Appraoch
Similar to Cytolon's Patients as Partners Appraoch (20)
You can transfer your health data to another personal health record 

You can transfer your health data to another personal health record
Tools and Technology for Advancing Rare Disease Research and Drug Development

Tools and Technology for Advancing Rare Disease Research and Drug Development
Digital Health Technology: The Ultimate Patient Advocate

Digital Health Technology: The Ultimate Patient Advocate
Identifying and Engaging Risk Stratified Patient Populations

Identifying and Engaging Risk Stratified Patient Populations
introductoin to Biostatistics ( 1st and 2nd lec ).ppt

introductoin to Biostatistics ( 1st and 2nd lec ).ppt
Providing additional information aboutthe benefits of statin.docx

Providing additional information aboutthe benefits of statin.docx
STANDARD OPERATING PROCEDURE OF CLINICAL PHARMACY DEPARTMENT

STANDARD OPERATING PROCEDURE OF CLINICAL PHARMACY DEPARTMENT
Note This assignment is for academic research pro only Thank y.docx

Note This assignment is for academic research pro only Thank y.docx
Piloting a Comprehensive Knowledge Base for Pharmacovigilance Using Standardi...

Piloting a Comprehensive Knowledge Base for Pharmacovigilance Using Standardi...
Recently uploaded
Contact dermaititis (irritant and allergic).pdf

Contact dermaititis (irritant and allergic).pdfChulalongkorn Allergy and Clinical Immunology Research Group
Recently uploaded (20)
TEST BANK For Williams' Essentials of Nutrition and Diet Therapy, 13th Editio...

TEST BANK For Williams' Essentials of Nutrition and Diet Therapy, 13th Editio...
New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...

New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...
Is preeclampsia and spontaneous preterm delivery associate with vascular and ...

Is preeclampsia and spontaneous preterm delivery associate with vascular and ...
Couples presenting to the infertility clinic- Do they really have infertility...

Couples presenting to the infertility clinic- Do they really have infertility...
ANATOMY OF THE LOWER URINARY TRACT AND MALE [Autosaved] [Autosaved].pptx![ANATOMY OF THE LOWER URINARY TRACT AND MALE [Autosaved] [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![ANATOMY OF THE LOWER URINARY TRACT AND MALE [Autosaved] [Autosaved].pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
ANATOMY OF THE LOWER URINARY TRACT AND MALE [Autosaved] [Autosaved].pptx
How to Give Better Lectures: Some Tips for Doctors

How to Give Better Lectures: Some Tips for Doctors
"Central Hypertension"‚ in China: Towards the nation-wide use of SphygmoCor t...

"Central Hypertension"‚ in China: Towards the nation-wide use of SphygmoCor t...
Relationship between vascular system disfunction, neurofluid flow and Alzheim...

Relationship between vascular system disfunction, neurofluid flow and Alzheim...
Why invest into infodemic management in health emergencies

Why invest into infodemic management in health emergencies
Anuman- An inference for helpful in diagnosis and treatment

Anuman- An inference for helpful in diagnosis and treatment
TEST BANK For Advanced Practice Nursing in the Care of Older Adults, 2nd Edit...

TEST BANK For Advanced Practice Nursing in the Care of Older Adults, 2nd Edit...
Factors Affecting child behavior in Pediatric Dentistry

Factors Affecting child behavior in Pediatric Dentistry
Non-Invasive assessment of arterial stiffness in advanced heart failure patie...

Non-Invasive assessment of arterial stiffness in advanced heart failure patie...
Blue Printing in medical education by Dr.Mumtaz Ali.pptx

Blue Printing in medical education by Dr.Mumtaz Ali.pptx
Cytolon's Patients as Partners Appraoch
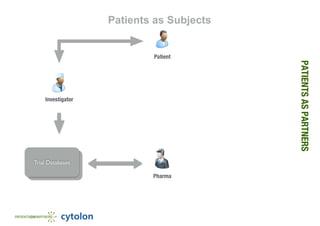
- 1. PATIENTSASPARTNERS Patients as Subjects Pharma Trial DatabasesTrial Databases Patient Investigator
- 2. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Investigator ?
- 3. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Investigator Peer communication before, during, and after clinical trial
- 4. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Investigator Individual trial data and human-readable summaries Peer communication before, during, and after clinical trial
- 5. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Investigator Primary care physician Data sharing Data sharing Individual trial data and human-readable summaries Peer communication before, during, and after clinical trial
- 6. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Investigator Primary care physician Data sharing Data sharing Individual trial data and human-readable summaries Peer communication before, during, and after clinical trial
- 7. PATIENTSASPARTNERS Patients as Partners Pharma Trial DatabasesTrial Databases Patient Researchers Investigator Primary care physician Data donation (de-identified) Data sharing Integration of patient-reported outcomes Data sharing Patient is the sole owner of his/her data Individual trial data and human-readable summaries Peer communication before, during, and after clinical trial
