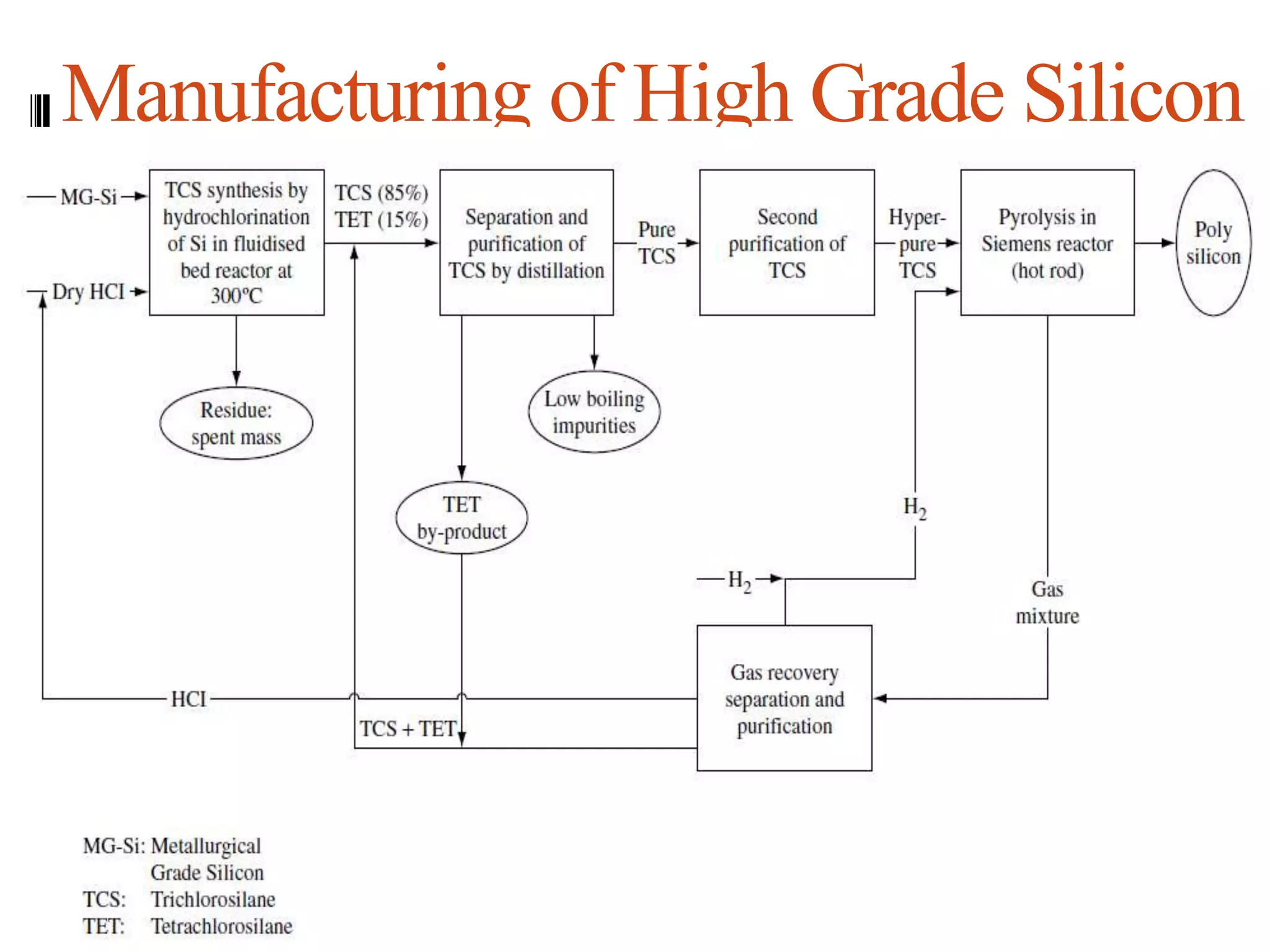
The document summarizes the process for producing high-grade silicon. It involves thermally decomposing trichlorosilane (SiHCl3) at 1100°C in a Siemens reactor (Siemens process). Feedstock is metallurgical-grade silicon. The process includes hydrochlorination to synthesize trichlorosilane, pyrolysis in a Siemens reactor to produce silicon and hydrogen chloride, and separation of trichlorosilane from byproducts using distillation. The purified trichlorosilane undergoes further distillation for higher purity. Recycling of byproducts occurs. The Siemens process produces high purity silicon widely used in semiconductors.