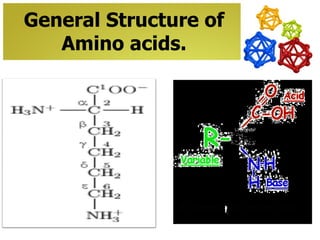
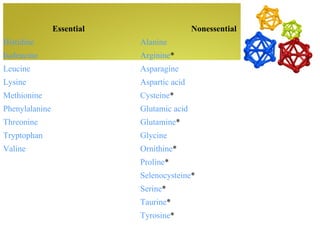
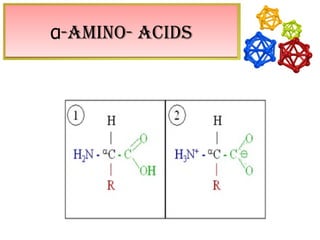
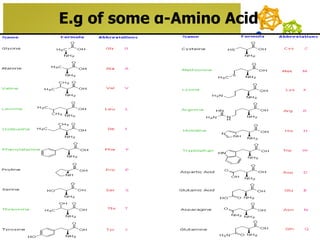
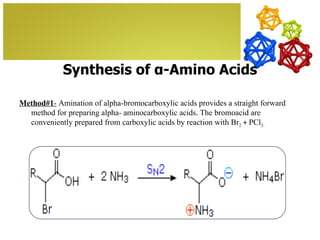
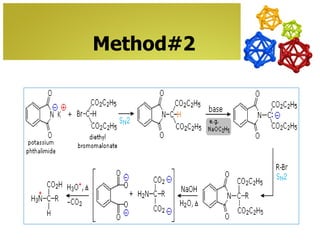
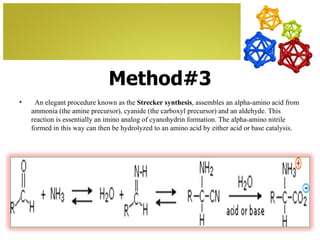
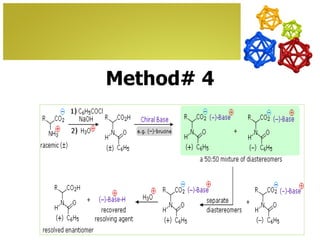
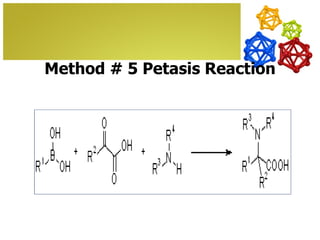
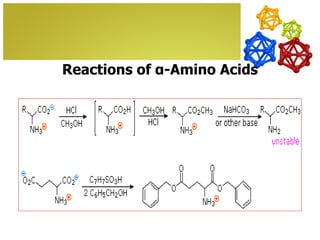
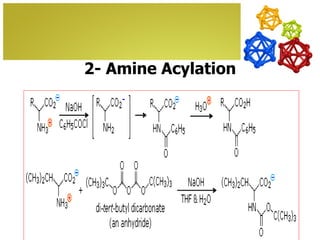
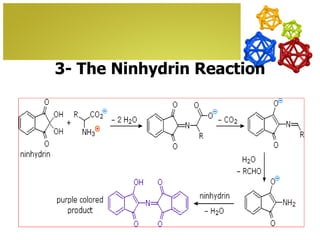
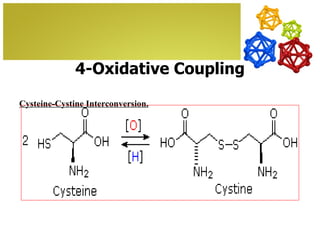
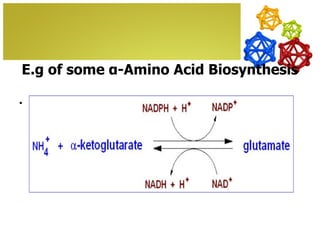
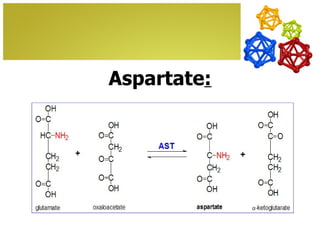
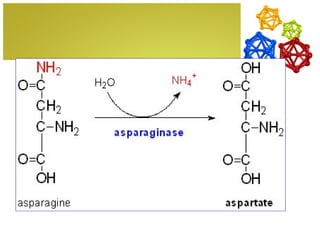
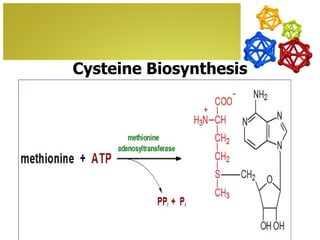
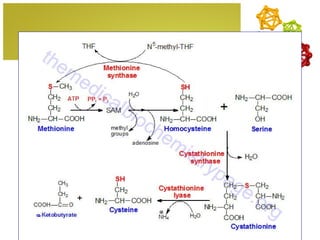
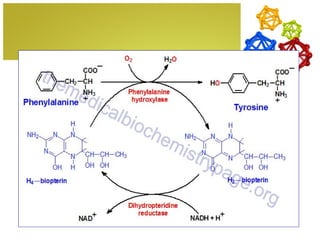
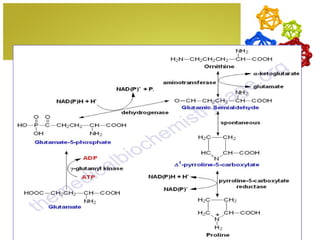
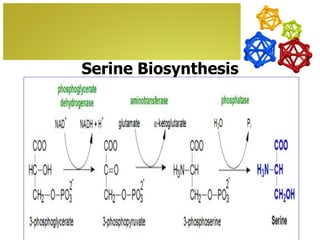
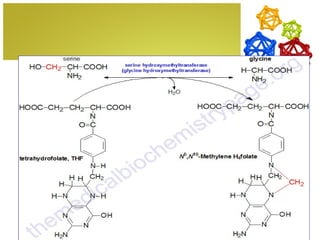
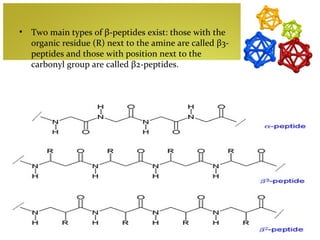
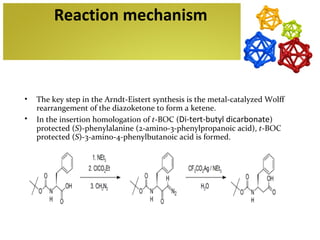
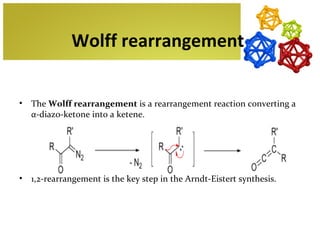
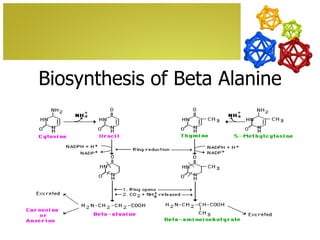
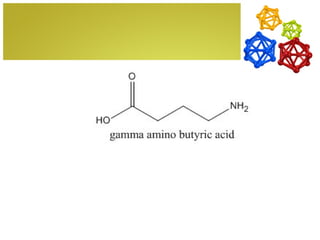
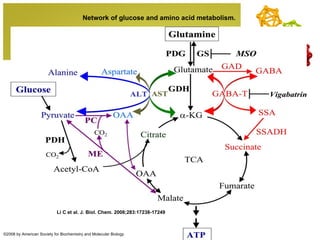
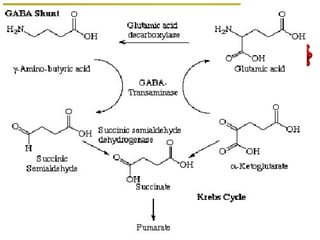
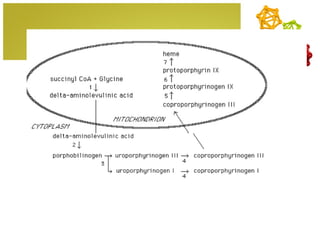
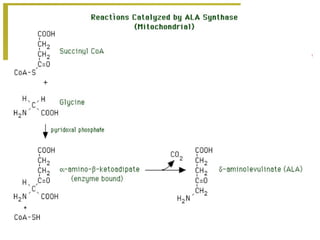
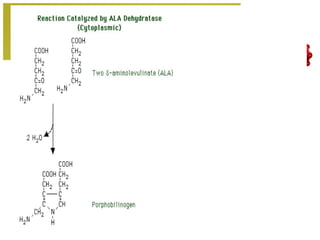
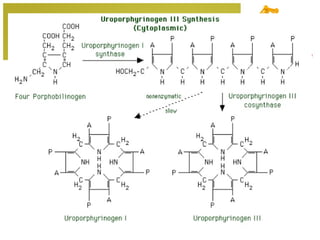
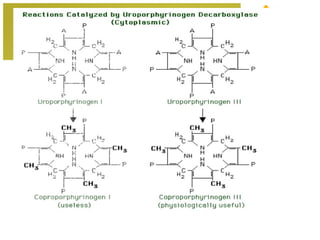
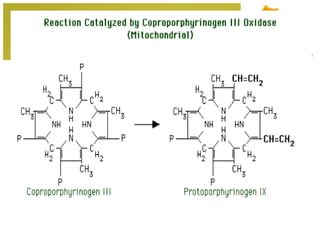
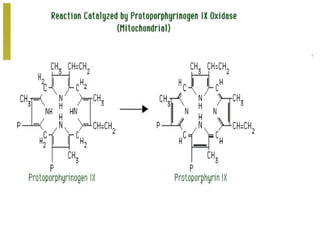
The document discusses amino acids, which are molecules that contain an amine group, a carboxylic acid group, and a side chain. There are 22 standard amino acids that are incorporated into proteins, as well as non-standard amino acids. Amino acids join together to form peptides or longer protein chains. Methods for synthesizing alpha-amino acids include amination reactions, Gabriel synthesis, Strecker synthesis, resolution of racemic mixtures, and the Petasis reaction. Amino acids undergo various reactions including esterification, acylation, and the ninhydrin reaction.