ψ(Wave function) and ψ2
•
0 likes•699 views
Significance of ψ(Wave function) and ψ2
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Particle in a box- Application of Schrodinger wave equation

Particle in a box- Application of Schrodinger wave equation
More from Mithil Fal Desai
More from Mithil Fal Desai (20)
How long could one survive in a perfectly airtight room.docx

How long could one survive in a perfectly airtight room.docx
Writing Third Year Project Report: Some Handy Guidelines

Writing Third Year Project Report: Some Handy Guidelines
Determine the strength in grams per litre of a given AgNO3 solution being pro...

Determine the strength in grams per litre of a given AgNO3 solution being pro...
Estimation of water of crystallization in mohr’s salt by titrating with stand...

Estimation of water of crystallization in mohr’s salt by titrating with stand...
Recently uploaded
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across SectorsAssociation for Project Management
Recently uploaded (20)
JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...

JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...

Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Separation of Lanthanides/ Lanthanides and Actinides

Separation of Lanthanides/ Lanthanides and Actinides
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across Sectors
ψ(Wave function) and ψ2
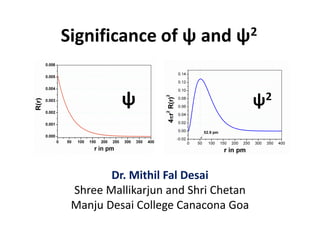
- 1. Significance of ψ and ψ2 Dr. Mithil Fal Desai Shree Mallikarjun and Shri Chetan Manju Desai College Canacona Goa ψ ψ2
- 2. Ψ and Ψ2 Ψ (Psi) is a wave function represented in the Schrodinger wave equation, which represents the state of the electron. Its corresponding probability density (Ψ2) gives the probability of finding the electron in a particular region in space.
- 4. Properties of wave function (ψ)
- 5. End note Several wave function satisfy these properties and each of these have corresponding energies. These wave functions are called orbitals. In hydrogen atom the wave function which has lowest energy level(E1) is 𝝍1. There are number of acceptable solutions of the wave function and each orbital is described by set of three quantum numbers n, l and m.