Quantum numbers
•Download as PPTX, PDF•
0 likes•43 views
This document describes the electron configuration of carbon. It shows that carbon has 6 protons and 6 electrons. The electrons fill the 1s, 2s, and 2p subshells according to the Aufbau principle and Hund's rule. Specifically, carbon has an electron configuration of 1s2 2s2 2p2.
Report
Share
Report
Share
Recommended
Recommended
The Author of this document is
Dr. Abdulfatah A. SalemOperations Management - Book1.p - Dr. Abdulfatah A. Salem

Operations Management - Book1.p - Dr. Abdulfatah A. SalemArab Academy for Science, Technology and Maritime Transport
https://app.box.com/s/cbgl8f0rgcll2fzdqp83sjxx8nom8188TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...Nguyen Thanh Tu Collection
https://app.box.com/s/z2cfx5b2yooxq1ov1wrd1dezn6af36uxBỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...

BỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...Nguyen Thanh Tu Collection
More Related Content
Recently uploaded
The Author of this document is
Dr. Abdulfatah A. SalemOperations Management - Book1.p - Dr. Abdulfatah A. Salem

Operations Management - Book1.p - Dr. Abdulfatah A. SalemArab Academy for Science, Technology and Maritime Transport
https://app.box.com/s/cbgl8f0rgcll2fzdqp83sjxx8nom8188TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...Nguyen Thanh Tu Collection
https://app.box.com/s/z2cfx5b2yooxq1ov1wrd1dezn6af36uxBỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...

BỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...Nguyen Thanh Tu Collection
Recently uploaded (20)
slides CapTechTalks Webinar May 2024 Alexander Perry.pptx

slides CapTechTalks Webinar May 2024 Alexander Perry.pptx
Operations Management - Book1.p - Dr. Abdulfatah A. Salem

Operations Management - Book1.p - Dr. Abdulfatah A. Salem
Post Exam Fun(da) Intra UEM General Quiz 2024 - Prelims q&a.pdf

Post Exam Fun(da) Intra UEM General Quiz 2024 - Prelims q&a.pdf
Post Exam Fun(da) Intra UEM General Quiz - Finals.pdf

Post Exam Fun(da) Intra UEM General Quiz - Finals.pdf
TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT VẬT LÝ 2024 - TỪ CÁC TRƯỜNG, TRƯ...
Pragya Champions Chalice 2024 Prelims & Finals Q/A set, General Quiz

Pragya Champions Chalice 2024 Prelims & Finals Q/A set, General Quiz
BỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...

BỘ LUYỆN NGHE TIẾNG ANH 8 GLOBAL SUCCESS CẢ NĂM (GỒM 12 UNITS, MỖI UNIT GỒM 3...
ppt your views.ppt your views of your college in your eyes

ppt your views.ppt your views of your college in your eyes
Featured
More than Just Lines on a Map: Best Practices for U.S Bike Routes
This session highlights best practices and lessons learned for U.S. Bike Route System designation, as well as how and why these routes should be integrated into bicycle planning at the local and regional level.
Presenters:
Presenter: Kevin Luecke Toole Design Group
Co-Presenter: Virginia Sullivan Adventure Cycling AssociationMore than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
Featured (20)
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...

Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike Routes
Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...

Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...
Good Stuff Happens in 1:1 Meetings: Why you need them and how to do them well

Good Stuff Happens in 1:1 Meetings: Why you need them and how to do them well
Quantum numbers
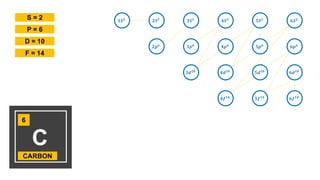
- 1. 𝟏𝑺 𝟐 𝟐𝑺 𝟐 𝟑𝑺 𝟐 𝟒𝑺 𝟐 𝟓𝑺 𝟐 𝟔𝑺 𝟐 𝟐𝒑 𝟔 𝟑𝒑 𝟔 𝟒𝒑 𝟔 𝟓𝒑 𝟔 𝟔𝒑 𝟔 𝟑𝒅 𝟏𝟎 𝟒𝒅 𝟏𝟎 𝟓𝒅 𝟏𝟎 𝟔𝒅 𝟏𝟎 𝟒𝒇 𝟏𝟒 𝟓𝒇 𝟏𝟒 𝟔𝒇 𝟏𝟒 6 C CARBON S = 2 P = 6 D = 10 F = 14
- 2. 𝟏𝑺 𝟐 𝟐𝑺 𝟐 𝟑𝑺 𝟐 𝟒𝑺 𝟐 𝟓𝑺 𝟐 𝟔𝑺 𝟐 𝟐𝒑 𝟔 𝟑𝒑 𝟔 𝟒𝒑 𝟔 𝟓𝒑 𝟔 𝟔𝒑 𝟔 𝟑𝒅 𝟏𝟎 𝟒𝒅 𝟏𝟎 𝟓𝒅 𝟏𝟎 𝟔𝒅 𝟏𝟎 𝟒𝒇 𝟏𝟒 𝟓𝒇 𝟏𝟒 𝟔𝒇 𝟏𝟒 6 C CARBON S = 2 P = 6 D = 10 F = 14 1𝑠2 2𝑠2 2𝑝4 2+2+4 = 6
- 3. PRINCIPLE QUANTUM NUMBER n = 1,3,4,5 Correspond to the energy level of the electrons
- 4. PRINCIPLE QUANTUM NUMBER n = 2 1𝑠2 2𝑠2 2𝑝4 6 C CARBON n Finding
- 5. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box
- 6. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 boxELECTRON SPIN
- 7. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 boxELECTRON SPIN
- 8. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 boxELECTRON SPIN
- 9. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 boxELECTRON SPIN
- 10. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ml Finding
- 11. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ml Finding -1
- 12. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ml Finding 0
- 13. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ml Finding 1
- 14. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ml Finding -1
- 15. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ms Finding +1/2
- 16. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ms Finding +1/2
- 17. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ms Finding +1/2
- 18. ANGULAR MOMENTUM S = 0 P = 1 D = 2 F = 3 This number defines the types of subshell 1𝑠2 2𝑠2 2𝑝46 C CARBON l Finding 1 box 3 box 5 box 7 box 0 1-1 ms Finding −1/2
- 19. 6 n CARBON 1𝑠2 2𝑠2 2𝑝4 ELECTRON CONFIGURATION l ml ms 2 2 2 2 1 1 1 1 -1 0 1 -1 +1/2 +1/2 +1/2 −1/2 HOW ? HOW ? HOW ? HOW ? HOW ? C
