Pt40 Propsdefined Reg
•Download as PPTX, PDF•
1 like•191 views
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (19)
Viewers also liked
Viewers also liked (12)
Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES

Chemistry(class11)-CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
Classification of elements and periodicity in properties class 11th chapter 3...

Classification of elements and periodicity in properties class 11th chapter 3...
Inorganic Chemistry : Periodic Table (Periodicity)

Inorganic Chemistry : Periodic Table (Periodicity)
IB Chemistry on Atomic Structure, Particle Physics and Relative Atomic Mass

IB Chemistry on Atomic Structure, Particle Physics and Relative Atomic Mass
Similar to Pt40 Propsdefined Reg
Similar to Pt40 Propsdefined Reg (20)
Covalent_Bonding_Presentation_-_Revised_for_Webpage.ppt

Covalent_Bonding_Presentation_-_Revised_for_Webpage.ppt
Recently uploaded
Driving Behavioral Change for Information Management through Data-Driven Gree...

Driving Behavioral Change for Information Management through Data-Driven Gree...Enterprise Knowledge
Recently uploaded (20)
Handwritten Text Recognition for manuscripts and early printed texts

Handwritten Text Recognition for manuscripts and early printed texts
Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...

Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...
08448380779 Call Girls In Greater Kailash - I Women Seeking Men

08448380779 Call Girls In Greater Kailash - I Women Seeking Men
The Codex of Business Writing Software for Real-World Solutions 2.pptx

The Codex of Business Writing Software for Real-World Solutions 2.pptx
The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf

The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf
Breaking the Kubernetes Kill Chain: Host Path Mount

Breaking the Kubernetes Kill Chain: Host Path Mount
How to Troubleshoot Apps for the Modern Connected Worker

How to Troubleshoot Apps for the Modern Connected Worker
WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service

WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service
Driving Behavioral Change for Information Management through Data-Driven Gree...

Driving Behavioral Change for Information Management through Data-Driven Gree...
Boost PC performance: How more available memory can improve productivity

Boost PC performance: How more available memory can improve productivity
IAC 2024 - IA Fast Track to Search Focused AI Solutions

IAC 2024 - IA Fast Track to Search Focused AI Solutions
Neo4j - How KGs are shaping the future of Generative AI at AWS Summit London ...

Neo4j - How KGs are shaping the future of Generative AI at AWS Summit London ...
Tata AIG General Insurance Company - Insurer Innovation Award 2024

Tata AIG General Insurance Company - Insurer Innovation Award 2024
Pt40 Propsdefined Reg
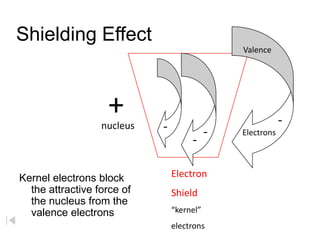
- 1. Shielding Effect Valence + - - nucleus - Electrons - Electron Shield “kernel” electrons Kernel electrons block the attractive force of the nucleus from the valence electrons
- 3. Periodic Trends in Atomic Radii LeMay Jr, Beall, Robblee, Brower, Chemistry Connections to Our Changing World , 1996, page 175
- 4. ELECTRONEGATIVITY—a measure of how strongly an atom’s nucleus pulls electron density toward itself when in a bond. *assigned (Pauling scale) *only useful in comparing two atoms (MEANS THAT ALL YOU CARE ABOUT IS THE DIFFERENCE!!!!)
- 5. Which has greater electronegativity in each compound?
- 7. the more tightly an electron is held, the more energy is required