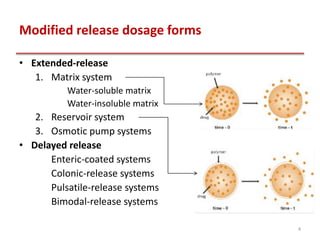
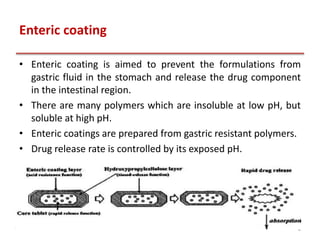
This document provides an overview of enteric coating polymers that are used to protect acid-labile drugs and ensure optimal drug absorption. It discusses various categories of enteric coating polymers including polymethacrylates (Eudragit), cellulose esters, and polyvinyl derivatives. Key points include: Eudragit polymers are commonly used methacrylic acid copolymers that are insoluble in gastric fluid but dissolve in the intestine. Cellulose esters like cellulose acetate phthalate are also widely employed. These polymers form films that protect the drug core from gastric conditions and dissolve above pH 6, allowing drug release in the intestines. The solubility and properties of different enteric coating polymers allow controlling