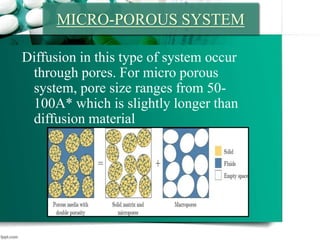
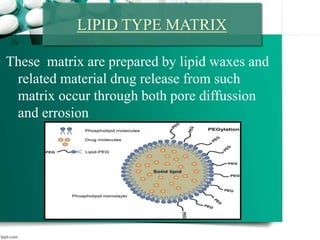
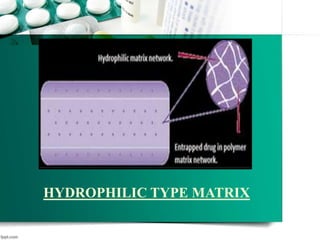
This document discusses various topics related to pharmaceutical technology and polymerization, including:
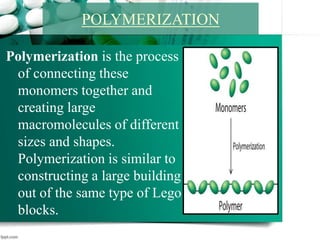
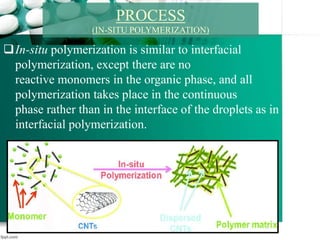
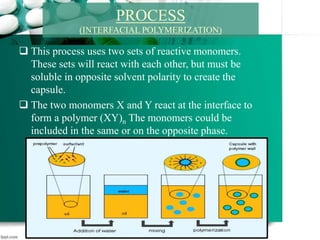
1. It introduces interfacial polymerization and microencapsulation using two types of polymerization - in-situ and interfacial polymerization.
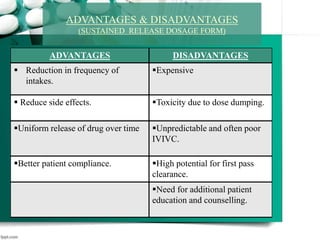
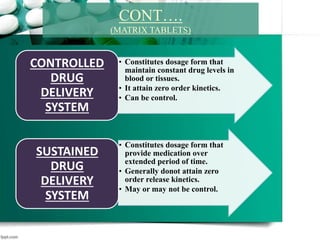
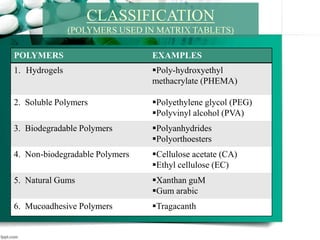
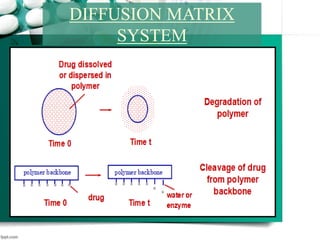
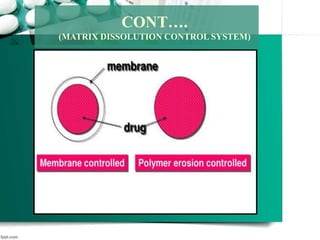
2. It then discusses sustained release drug delivery systems, highlighting matrix tablets as a common approach and factors that influence drug release.
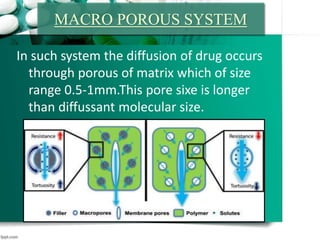
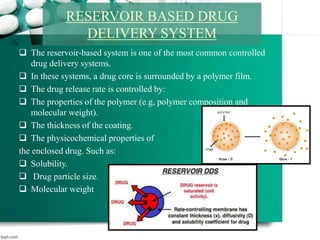
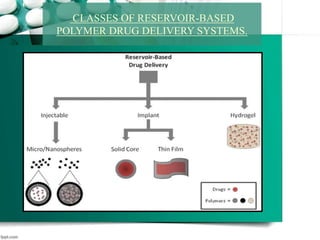
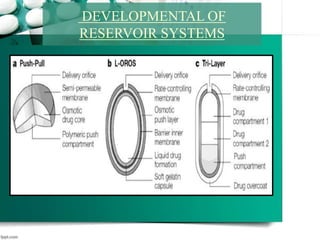
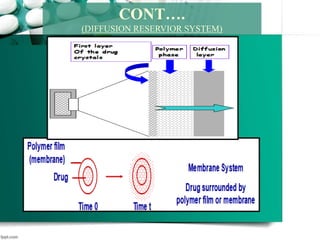
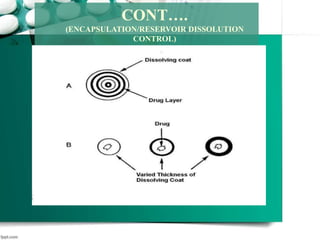
3. Finally, it covers reservoir-based drug delivery systems, describing benefits, classes, development approaches, and common polymers used including non-biodegradable and biodegradable options.