Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
APPLIED THERMODYNAMICS 18ME42 Module 03: Vapour Power Cycles

APPLIED THERMODYNAMICS 18ME42 Module 03: Vapour Power Cycles
Axial compressor - variation of rotor and stator angles from root to tip - 4t...

Axial compressor - variation of rotor and stator angles from root to tip - 4t...
Axial compressor theory - stage-wise isentropic efficiency - 18th March 2010

Axial compressor theory - stage-wise isentropic efficiency - 18th March 2010
Aircraft propulsion non ideal turbofan cycle analysis

Aircraft propulsion non ideal turbofan cycle analysis
I.C ENGINE LAB. Performance of The Four Strokes Diesel Engine

I.C ENGINE LAB. Performance of The Four Strokes Diesel Engine
Similar to Lect 5
Similar to Lect 5 (20)
RADIAL HEAT CONDUCTION SOLVED USING THE INTEGRAL EQUATION .pdf

RADIAL HEAT CONDUCTION SOLVED USING THE INTEGRAL EQUATION .pdf
Equations_3_Industrial Instrumentation - Temperature & Level Measurement Impo...

Equations_3_Industrial Instrumentation - Temperature & Level Measurement Impo...
Recently uploaded
Welcome to the April edition of WIPAC Monthly, the magazine brought to you by Water Industry Process Automation & Control.
In this month's edition, along with the latest news from the industry we have articles on:
The use of artificial intelligence and self-service platforms to improve water sustainability
A feature article on measuring wastewater spills
An article on the National Underground Asset Register
Have a good month,
OliverWater Industry Process Automation & Control Monthly - April 2024

Water Industry Process Automation & Control Monthly - April 2024Water Industry Process Automation & Control
Process of Integration the Laser Scan Data into FEA Model and Level 3 Fitness-for-Service Assessment of Critical Assets in Refinery & Process IndustriesFEA Based Level 3 Assessment of Deformed Tanks with Fluid Induced Loads

FEA Based Level 3 Assessment of Deformed Tanks with Fluid Induced LoadsArindam Chakraborty, Ph.D., P.E. (CA, TX)
Call girls in delhi ✔️✔️🔝 9953056974 🔝✔️✔️Welcome To Vip Escort Services In Delhi [ ]Noida Gurgaon 24/7 Open Sex Escort Services With Happy Ending ServiCe Done By Most Attractive Charming Soft Spoken Bold Beautiful Full Cooperative Independent Escort Girls ServiCe In All-Star Hotel And Home Service In All Over Delhi, Noida, Gurgaon, Faridabad, Ghaziabad, Greater Noida,
• IN CALL AND OUT CALL SERVICE IN DELHI NCR
• 3* 5* 7* HOTELS SERVICE IN DELHI NCR
• 24 HOURS AVAILABLE IN DELHI NCR
• INDIAN, RUSSIAN, PUNJABI, KASHMIRI ESCORTS
• REAL MODELS, COLLEGE GIRLS, HOUSE WIFE, ALSO AVAILABLE
• SHORT TIME AND FULL TIME SERVICE AVAILABLE
• HYGIENIC FULL AC NEAT AND CLEAN ROOMS AVAIL. IN HOTEL 24 HOURS
• DAILY NEW ESCORTS STAFF AVAILABLE
• MINIMUM TO MAXIMUM RANGE AVAILABLE.
Call Girls in Delhi & Independent Escort Service –
CALL GIRLS SERVICE DELHI NCR
Vip call girls in Delhi
Call Girls in Delhi, Call Girl Service 24×7 open
Call Girls in Delhi Best Delhi Escorts in Delhi
Low Rate Call Girls In Saket Delhi
X~CALL GIRLS IN Ramesh Nagar Metro
best Delhi call girls and Delhi escort service.
CALL GIRLS SERVICE IN ALL DELHI …
(Delhi) Call Girls in (Chanakyapuri)
Hot And Sexy Independent Model Escort Service In Delhi Unlimited Enjoy Genuine 100% Profiles And Trusted Door Step Call Girls Feel Free To Call Us Female Service Hot Busty & Sexy Party Girls Available For Complete Enjoyment. We Guarantee Full Satisfaction & In Case Of Any Unhappy Experience, We Would Refund Your Fees, Without Any Questions Asked. Feel Free To Call Us Female Service Provider Hours Opens Thanks.
Delhi Escorts Services 100% secure Services.Incall_OutCall Available and outcall Services provide.
We are available 24*7 for Full Night and short Time Escort Services all over Delhi NCR.
Delhi All Hotel Services available 3* 4* 5* Call Call
Delhi Escorts Services And Delhi Call Girl Agency 100% secure Services in my agency. Incall and outcall Services provide.
We are available 24*7 for Full Night and short Time Escort Services my agency in all over New Delhi
Delhi All Hotel Services available my agency
SERVICES [✓✓✓]
Housewife
College Girl
VIP Escort
Independent Girl
Aunty
Without a Condom sucking )?
Sexy Aunty.DSL (Dick Sucking Lips)?
DT (Dining at the Toes English Spanking)
Doggie (Sex style from no behind)??
OutCall- All Over Delhi Noida Gurgaon 24/7
FOR APPOINTMENT Call/Whatsop / 9953056974Call Girls in Netaji Nagar, Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service

Call Girls in Netaji Nagar, Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
From customer value engagements to hands-on production support, our Services span across every stage of our customers digital transformation journey, to help ensure that every customer is successful in their adoption of our solutions.
• Implementation, Upgrade, Migration, and Maintenance Services
• On-Premises and On-Cloud
• COTS Training Services; On-Site and Virtual
• Software Support Services; Legacy and 3DEXPERIENCE
• Value Engagement & Blueprinting
• Specialized Consulting and Support Services
• Customized Training Services
• Automation and Configuration Services
• Technical Resource Augmentation Services
• Project Management
• Know-how Training (mentoring) and Resource AugmentationNavigating Complexity: The Role of Trusted Partners and VIAS3D in Dassault Sy...

Navigating Complexity: The Role of Trusted Partners and VIAS3D in Dassault Sy...Arindam Chakraborty, Ph.D., P.E. (CA, TX)
Call girls in delhi ✔️✔️🔝 9953056974 🔝✔️✔️Welcome To Vip Escort Services In Delhi [ ]Noida Gurgaon 24/7 Open Sex Escort Services With Happy Ending ServiCe Done By Most Attractive Charming Soft Spoken Bold Beautiful Full Cooperative Independent Escort Girls ServiCe In All-Star Hotel And Home Service In All Over Delhi, Noida, Gurgaon, Faridabad, Ghaziabad, Greater Noida,
• IN CALL AND OUT CALL SERVICE IN DELHI NCR
• 3* 5* 7* HOTELS SERVICE IN DELHI NCR
• 24 HOURS AVAILABLE IN DELHI NCR
• INDIAN, RUSSIAN, PUNJABI, KASHMIRI ESCORTS
• REAL MODELS, COLLEGE GIRLS, HOUSE WIFE, ALSO AVAILABLE
• SHORT TIME AND FULL TIME SERVICE AVAILABLE
• HYGIENIC FULL AC NEAT AND CLEAN ROOMS AVAIL. IN HOTEL 24 HOURS
• DAILY NEW ESCORTS STAFF AVAILABLE
• MINIMUM TO MAXIMUM RANGE AVAILABLE.
Call Girls in Delhi & Independent Escort Service –
CALL GIRLS SERVICE DELHI NCR
Vip call girls in Delhi
Call Girls in Delhi, Call Girl Service 24×7 open
Call Girls in Delhi Best Delhi Escorts in Delhi
Low Rate Call Girls In Saket Delhi
X~CALL GIRLS IN Ramesh Nagar Metro
best Delhi call girls and Delhi escort service.
CALL GIRLS SERVICE IN ALL DELHI …
(Delhi) Call Girls in (Chanakyapuri)
Hot And Sexy Independent Model Escort Service In Delhi Unlimited Enjoy Genuine 100% Profiles And Trusted Door Step Call Girls Feel Free To Call Us Female Service Hot Busty & Sexy Party Girls Available For Complete Enjoyment. We Guarantee Full Satisfaction & In Case Of Any Unhappy Experience, We Would Refund Your Fees, Without Any Questions Asked. Feel Free To Call Us Female Service Provider Hours Opens Thanks.
Delhi Escorts Services 100% secure Services.Incall_OutCall Available and outcall Services provide.
We are available 24*7 for Full Night and short Time Escort Services all over Delhi NCR.
Delhi All Hotel Services available 3* 4* 5* Call Call
Delhi Escorts Services And Delhi Call Girl Agency 100% secure Services in my agency. Incall and outcall Services provide.
We are available 24*7 for Full Night and short Time Escort Services my agency in all over New Delhi
Delhi All Hotel Services available my agency
SERVICES [✓✓✓]
Housewife
College Girl
VIP Escort
Independent Girl
Aunty
Without a Condom sucking )?
Sexy Aunty.DSL (Dick Sucking Lips)?
DT (Dining at the Toes English Spanking)
Doggie (Sex style from no behind)??
OutCall- All Over Delhi Noida Gurgaon 24/7
FOR APPOINTMENT Call/Whatsop / 9953056974Call Girls in Ramesh Nagar Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service

Call Girls in Ramesh Nagar Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Double Revolving field theory-how the rotor develops torque

Double Revolving field theory-how the rotor develops torque
Water Industry Process Automation & Control Monthly - April 2024

Water Industry Process Automation & Control Monthly - April 2024
VIP Call Girls Palanpur 7001035870 Whatsapp Number, 24/07 Booking

VIP Call Girls Palanpur 7001035870 Whatsapp Number, 24/07 Booking
FEA Based Level 3 Assessment of Deformed Tanks with Fluid Induced Loads

FEA Based Level 3 Assessment of Deformed Tanks with Fluid Induced Loads
Call Girls in Netaji Nagar, Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service

Call Girls in Netaji Nagar, Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service
Design For Accessibility: Getting it right from the start

Design For Accessibility: Getting it right from the start
Call Girls Pimpri Chinchwad Call Me 7737669865 Budget Friendly No Advance Boo...

Call Girls Pimpri Chinchwad Call Me 7737669865 Budget Friendly No Advance Boo...
XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX

XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX
Navigating Complexity: The Role of Trusted Partners and VIAS3D in Dassault Sy...

Navigating Complexity: The Role of Trusted Partners and VIAS3D in Dassault Sy...
Call Girls Wakad Call Me 7737669865 Budget Friendly No Advance Booking

Call Girls Wakad Call Me 7737669865 Budget Friendly No Advance Booking
Call Girls in Ramesh Nagar Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service

Call Girls in Ramesh Nagar Delhi 💯 Call Us 🔝9953056974 🔝 Escort Service
VIP Call Girls Ankleshwar 7001035870 Whatsapp Number, 24/07 Booking

VIP Call Girls Ankleshwar 7001035870 Whatsapp Number, 24/07 Booking
Lect 5
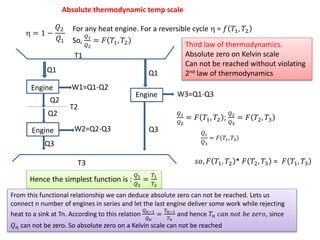
- 1. Absolute thermodynamic temp scale = 1 − 𝑄2 𝑄1 For any heat engine. For a reversible cycle = 𝑓 𝑇1, 𝑇2 So, 𝑄1 𝑄2 = 𝐹 𝑇1, 𝑇2 T1 T2 Q1 Q2 Q2 Q3 T3 W1=Q1-Q2 W2=Q2-Q3 Q1 Q3 W3=Q1-Q3 𝑄1 𝑄2 = 𝐹 𝑇1, 𝑇2 ; 𝑄2 𝑄3 = 𝐹 𝑇2, 𝑇3 𝑄1 𝑄3 = 𝐹 𝑇1, 𝑇3 𝑠𝑜, 𝐹 𝑇1, 𝑇2 * 𝐹 𝑇2, 𝑇3 = 𝐹 𝑇1, 𝑇3 Hence the simplest function is : 𝑄1 𝑄2 = 𝑇1 𝑇2 From this functional relationship we can deduce absolute zero can not be reached. Lets us connect n number of engines in series and let the last engine deliver some work while rejecting heat to a sink at Tn. According to this relation 𝑄 𝑛−1 𝑄 𝑛 = 𝑇 𝑛−1 𝑇 𝑛 and hence 𝑇𝑛 𝑐𝑎𝑛 𝑛𝑜𝑡 𝑏𝑒 𝑧𝑒𝑟𝑜, since 𝑄 𝑛 can not be zero. So absolute zero on a Kelvin scale can not be reached Third law of thermodynamics. Absolute zero on Kelvin scale Can not be reached without violating 2nd law of thermodynamics Engine Engine Engine
- 2. = 1 − 𝑄2 𝑄1 = 1 − 𝑇2 𝑇1 Efficiency of reversible heat engine 𝑐𝑜𝑝 ref = Q2 Q1 − Q2 = T2 T1 − T2 𝑐𝑜𝑝 HP = Q1 Q1 − Q2 = T1 T1 − T2 Internally reversible and externally irreversible process TH TL QH QL Ta Tb Engine W 𝑄 𝐻 = 𝑐 𝐻(𝑇 𝐻 − 𝑇𝑎) 𝑄 𝐿 = 𝑐 𝐿(𝑇𝑏 − 𝑇𝐿) 𝑊 = 𝑄 𝐻 − 𝑄 𝐿 If 𝑇 𝐻 ≈ 𝑇𝑎 and 𝑇𝑏 = 𝑇𝐿 then is max But QH=0 and w=0 On the other hand if 𝑇𝑎 ≪ 𝑇 𝐻, 𝑎𝑛𝑑 𝑇𝑏 ≫ 𝑇𝐿 𝑄 𝐻 ≫ 0, ≅ 0, 𝑎𝑛𝑑 𝑤 ≅ 0 So there is some optimal set of cycle temp for maximum power output. Find optimum 𝑇𝑎 𝑎𝑛𝑑 𝑇𝑏for max power. Known are: 𝐶 𝐻, 𝐶𝐿, 𝑇 𝐻, 𝑇𝐿 𝑎𝑛𝑑 𝑢𝑛𝑘𝑛𝑜𝑤𝑛 𝑎𝑟𝑒 𝑇𝑎 𝑎𝑛𝑑 𝑇𝑏, find max W ch=100; cl=80; th=900; tl=300 qh=ch*(th-ta) ql=cl*(tb-tl) w=qh-ql qh/ta=ql/tb ta_theo=ch/(ch+cl)*th+cl/(ch+cl)*sqrt(th*tl) tb_theo=cl/(ch+cl)*tl+ch/(ch+cl)*sqrt(th*tl) Make a parametric table with Ta, Tb, Qh, QL, and w Vary Ta from 900 to 630K and get the table solved. Now you can see W as a function of Ta and Tb. Find W_max from the table.