TOF.pptx
- 2. Most common cyanotic heart defect seen in children beyond infancy, accounting for a third of all congenital heart disease (CHD) in this age group
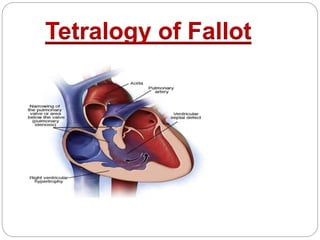
- 3. Tetralogy of Fallot (TOF) is a congenital heart defect, which has four anatomical components: Anterior malalignment ventricular septal defect (VSD) Aortic override over the muscular septum Variable degrees of subvalvar, valvar, and supravalvar pulmonary stenosis Right ventricular (RV) infundibular narrowing and RV hypertrophy
- 5. The four salient anatomic components of Fallot’s tetralogy result from a specific morphogenetic abnormality: Malalignment of the infundibular septum
- 7. Embryology In the normal heart, division of the fetal conotruncus culminates in alignment of the infundibular septum with the muscular trabecular septum In Fallot’s tetralogy, the infundibular septum deviates anteriorly and cephalad and is therefore not aligned with the trabecular septum, creating a ventricular septal defect at the site of malalignment The deviation of the infundibular septum encroaches on the right ventricular outflow tract and causes infundibular stenosis and a biventricular (overriding) aorta
- 9. Malaligned ventricular septal defects are located in the perimembranous septum with extension into the infundibular septum Atrioventricular conduction is normal
- 10. Ventricular septal defect This condition is usually a large non-restrictive defect (i.e. no obstruction to flow across the VSD) It is often located in the perimembranous and muscular regions of the ventricular septum, allowing shunting of blood between the ventricles (right to left in a classical cyanotic ToF)
- 11. RV outflow tract obstruction The obstruction to pulmonary blood flow at the level of the RV outflow tract (RVOT) is a key feature of ToF The RVOTO subsequently causes the RVH (another key feature of ToF) In the presence of an unrestrictive VSD, worsening RVOTO increases the RV pressure, drives a right-to-left shunt through the VSD, reduces pulmonary blood flow, and leads to hypoxaemia
- 12. Assuming the patent ductus arteriosus (PDA) is closed and there is no collateral supply, then the more severe the RVOTO, the more hypoxaemic the patient may be on presentation A key feature is obstruction at the sub-valvular RVOT (in 50% of patients) However, the obstruction may be at the pulmonary valve (10%), above the pulmonary valve (10%), or a mixture (30%) Complete occlusion of the RVOT is known as Pulmonary Atresia
- 13. Overriding aorta This condition describes the ventriculoarterial connection where the aorta can override the VSD to varying degrees, because of malalignment of the outlet component of the septum In ToF, the aorta still arises from the left ventricle, with only some origin from the right ventricle If the aorta arises predominantly from the right ventricle, then this abnormality may be called double- outlet right ventricle (DORV), and the physiology is determined by the VSD position and associated RV or left ventricular (LV ) outflow obstruction
- 15. RV hypertrophy RV hypertrophy develops as a consequence of the RVOTO because increased RV pressure needs to be generated to maintain pulmonary blood flow
- 16. Genetics Chromosomal anomalies are involved in about 12 percent of the cases, e.g. trisomy 21 (Down syndrome), trisomy 13 (Patau syndrome) and trisomy 18 (Edwards syndrome) DiGeorge Syndrome 22q11 microdeletion Alagille syndrome CHARGE (coloboma, heart defect, atresia choanae, retarded growth and development, genital abnormality, ear abnormality) syndrome VACTERL
- 17. Patients with TOF and Down syndrome frequently have a particularly large VSD in the inlet septum
- 19. Coronary abnormalities occur in 5 percent, anomalous origin of left anterior descending (LAD) artery from the right coronary artery (RCA) being the most common
- 20. Natural History Two thirds of patients reached their first birthday Approximately half reached age 3 years Approximately a quarter completed the first decade of life The attrition rate was then 6.4% per year with 11% alive at age 20 years 6% at age 30 years 3% at age 40 years
- 23. Natural History of Pulmonary Atresia If pulmonary atresia is present as well, survival is even poorer with only 50 percent of patients surviving to 1 year and only 8 percent of patients surviving to 10 years Adequate collateral blood flow occasionally permits survival into adolescence and adulthood
- 24. Hemodynamics
- 25. The important features that determine the hemodynamic consequences in patients with ventricular septal defect and pulmonary stenosis are the Size of the defect The severity of the stenosis, and Level of systemic vascular resistance
- 27. ‘Pink’ Fallot Children with this variant are acyanotic with normal or near-normal oxygen saturations There is minimal or no RVOTO Physiologically, the lesion behaves like a large unrestricted VSD with a left-to-right shunt Patients can present with heart failure or other features of the left-to-right shunt, although this condition can change with time as the child grows, and features are more like the features of a classical ToF
- 28. Fallot-type pulmonary atresia (15% of ToF) This type is the severest variant, characterised by complete atresia (i.e. complete occlusion) of the pulmonary valve Therefore, there is no forward flow of blood from the RV into the pulmonary artery Intracardiac mixing is essential, and all pulmonary blood flow must be supplied from the aorta either by a PDA (‘duct-dependent pulmonary circulation’) or from major collaterals from the aorta to the pulmonary arteries (major aortopulmonary collateral arteries, MAPCAs) In the neonatal period, prostaglandin (either alprostadil [prostaglandin E1] or dinoprostone [prostaglandin E2]) may be required acutely to maintain any pulmonary blood flow through the PDA
- 29. ToF with absent pulmonary valve (6% of ToF) There is no pulmonary valve, but the RVOT is open These infants are often acyanotic because there is no RVOTO, but the condition is notable for respiratory complications that develop secondary to massive aneurysmal dilatation of the pulmonary arteries caused by absence of the pulmonary valve, with obligatory pulmonary regurgitation These aneurysmal dilatations externally compress the distal trachea and bronchi causing intrathoracic airway obstruction, lung atelectasis,
- 31. The tetralogy usually comes to light in neonates and infants When the shunt is left-to-right, initial suspicion is a prominent systolic murmur When the shunt is balanced, the murmur persists in addition to mild, intermittent, or stress-induced cyanosis When the shunt is reversed, the prominence of the systolic murmur is inversely proportional to the degree of cyanosis
- 32. The clinical course in early infancy is often benign Mild to moderate neonatal cyanosis tends to increase, but cyanosis may be delayed for months and is coupled with increased oxygen requirements of the growing infant rather than with progressive obstruction to right ventricular outflow Patients seldom remain acyanotic after the first few years of life, and by 5 to 8 years of age, most children are conspicuously cyanotic, with cyanosis closely coupled to the severity of pulmonary stenosis
- 33. The degree of right ventricular outflow tract obstruction (RVOTO) often correlates with the degree of cyanosis and the timing of presentation Thus patients with mild pulmonary obstruction present late, perhaps even in adulthood, the so- called “pink TOF”, while patients with severe obstruction may present soon after birth on closure of the ductus arteriosus In less severe cases, cyanosis is first noticed during crying
- 34. Cyanotic Spells
- 35. Spell usually occurs in infants between 3 to 24 months of age Typical spell is characterized by progressive increase in the rate and depth of respiration, deepening cyanosis, limpness or syncope Convulsions, cerebrovasclur accident and death are potential complications Spells are less common after 2 years Initiated usually by crying, feeding or bowel movement, spells are particularly common after getting up from sleep
- 37. Wood's theory Hypoxemic spells are caused by: Spasm of the infundibulum of the right ventricle Progressively increasing right to left shunting Metabolic acidosis
- 38. Catecholamine release Leads to increased myocardial contractility and infundibular stenosis (both these theories do not explain the cause of cyanotic spells in patients with tetralogy of Fallot with pulmonary atresia).
- 39. Guntheroth's theory Episodes of paroxysmal hyperpnoea are the cause rather than the effect of cyanotic spells Hyperpnoea Increased systemic venous return Right to left shunt as well as oxygen consumption through increase work of breathing
- 40. Kothari's theory Stimulation of mechanoreceptors in the right ventricle to be the cause of spells
- 41. Morgan’s theory Vulnerable respiratory centre which over- reacts to hypoxic stimuli like crying, feeding
- 42. Young’s theory It was proposed that the spell was precipitated by an atrial tachycardia
- 44. Squatting in TOF
- 45. Squatting is of diagnostic significance in TOF Squatting increases peripheral vascular resistance and thus decreases the magnitude of the right to left shunt across the VSD Locking up the more desaturated lower limb venous blood and displacing the better oxygenated mesenteric venous blood into the right heart may be the other benefits of squatting
- 49. Recurrent hypoxic spells sometimes lead to brain damage and mental retardation Cerebral venous sinus thromboses and small occult thromboses may become manifest after prolonged hypoxic spells
- 50. Hypernasal resonance or nasal speech (velopharyngeal insufficiency) may develop after repeated or prolonged spells because nasal resonance is compromised by improper approximation of the velum (soft palate) and the pharyngeal walls Brain abscess and cerebral embolism add to the list of central nervous system complications
- 51. Iron deficient erythrocytosis in patients less than 4 years of age increases the risk of cerebral venous sinus thrombosis Wheezing and stridor have been attributed to tracheal compression by an enlarged aorta A stenotic pulmonary valve and an incompetent aortic valve are substrates for infective endocarditis
- 53. Most infants are smaller than expected for age Cyanosis of the lips and nail bed may be noticed at birth or may appear later Cyanosis in TOF is determined by the severity of pulmonary stenosis and also to a lesser extent by systemic to pulmonary collaterals Infundibular stenosis worsens as the infant grows so that a previously pink baby turns blue In the case of pulmonary atresia, cyanosis sometimes may be absent due to systemic pulmonary collaterals Clubbing may be present after 3 months of life.
- 54. S1 is normal, while S2 is single due to a faint P2 Delayed and hesitant closure of the pulmonary valve due to the slow pressure drop in the stenotic infundibular chamber, associated valvar stenosis and the overriding aorta all contribute to the single S2
- 55. A prominent ejection systolic murmur, is heard at the mid and upper left sternal border The intensity of this murmur is inversely proportional to the severity of stenosis With more severe stenosis RV pumps more into the aorta and less across the RVOT, decreasing the murmur The murmur disappears during a spell
- 56. An aortic ejection click due to the dilated ascending aorta may be heard over the apex A continuous murmur below the left clavicle denotes a patent ductus arteriosus (PDA) A more widely heard continuous murmur, especially over the back, is due to systemic- pulmonary collaterals
- 57. Investigations
- 58. ECG in TOF In the newborn, the EKG may be normal but over the first weeks of life normal regression of right ventricular preponderance is not seen Right ventricular hypertrophy is the hallmark EKG finding in the patient with tetralogy of Fallot and is of value in the differential diagnosis from ventricular septal defect Tall R waves in the right precordial leads (V1-V2) are usually seen
- 60. Whereas the R wave in V1 is tall and usually monophasic, R wave in V2 is much shorter – the so called “sudden transition” is characteristic
- 61. Right-axis deviation may accompany right ventricular hypertrophy, additionally, right atrial enlargement is manifested by tall P waves (P pulmonale) Left-axis deviation suggests an associated complete AV canal defect Sudden transition of the QRS complex morphology in leads V1 and V2 is a common pattern in patients with tetralogy of Fallot. There is a Rs pattern with a tall R wave in lead V1, and a rS pattern from leads V2 or V3 to lead V6
- 62. With pulmonary atresia and an abundant collateral arterial circulation, P waves are broad and bifid because of increased flow into the left atrium Q waves with well-developed R waves appear in leads V5-6 because of increased flow into the left ventricle ST segment and T wave abnormalities may be found in midprecordial leads
- 63. Chest Xray Findings on an x-ray diagnostic of tetralogy of fallot include: Normal or decreased pulmonary vascularity Concave pulmonary artery segment A right sided aortic arch may also be present. There is pruning or reduction in the prominence of the pulmonary vasculature over time
- 64. Chest X-Ray Plain films may classically show a "boot shaped" heart with an upturned cardiac apex due to right ventricular hypertrophy and concave pulmonary arterial segment
- 66. -The cardiac apex elevated suggestive of right ventricular enlargement -The main pulmonary artery segment is concave - Right-sided aortic arch is demonstrated - There is decreased pulmonary vascularity (pulmonary oligemia)
- 68. Parasternal long-axis view Left and right ventricular size/function Degree of aortic override override of the aortic root over the ventricular septal defect should be less than half of the aortic diameter Analysis of the ventricular septal defect magnitude and direction of shunting across the VSD Confirmation of aorto-mitral continuity absence of the fibrous continuity between the aortic and mitral valves is inconsistent with tetralogy of fallot, and may be suggestive of double outlet right ventricle (DORV)
- 69. Parasternal short-axis view Location of the ventricular septal defect Anatomy of the right ventricular outflow tract Dilation of the RVOT may be observed Dynamic obstruction of the RVOT may be noted, resulting from incursion of the infundibulum into the outflow tract
- 70. Pulmonic valve leaflet number and degree of mobility dimensions of the annulus spectral Doppler interrogation for the presence and grading of associated pulmonic regurgitation or stenosis
- 71. Apical 4 chamber view Assess right and left ventricular size and function Right ventricular hypertrophy may be assessed by measurement of the free wall of the right ventricle
- 73. Cath Study in TOF
- 74. Cardiac catheterization may be necessary in few cases to further delineate The levels of right ventricular outflow obstruction Branch pulmonary artery stenosis or hypoplasia Coronary artery anatomy Presence of aortopulmonary collaterals, and Presence of additional VSD
- 75. The hemodynamic findings at catheterization typically reveal normal or only mildly elevated filling pressures The left and right ventricular systolic pressures are equal Pulmonary artery pressures are normal or low The degree of right-to-left shunting is best shown by the degree of systemic desaturation
- 76. Angiographic assessment should be geared towards the information that is needed; biplane angiography is ideal RV angiogram (anteroposterior [AP] cranial, left anterior oblique [LAO] view) shows simultaneous opacification of aorta and PA, RVOT obstruction and PA anatomy
- 78. An aortic root injection will usually provide adequate identification of the coronary arteries, although selective injections may occasionally be needed The arch and descending aorta may also be seen in this view and show a PDA or collateral vessels If collateral vessels are identified, selective injections are helpful to assess the areas of the pulmonary bed that they supply and whether they are the sole supply to these areas
- 79. The VSD is best seen from a left ventricular injection in a long axial oblique projection Multidetector computed tomography (MDCT) angiography is a safe and effective non-invasive technique to answer questions remaining after echocardiography in patients with TOF Major aortopulmonary collateral arteries (MAPCA) from all sources are best shown by this technique
- 80. Management
- 83. A cyanotic spell is usually self limiting and lasts less than 15-30 minutes But sometimes they can be prolonged and require emergency measures like:
- 84. 1. Hold the child in knee chest position This increases the SVR and decreases the desaturated systemic venous return
- 86. 2. Calm the child. The ideal sedative is morphine It causes respiratory centre suppression and sedation thereby reducing hyperpnea Decreases systemic venous return (venodilator) and relax the infundibulum The dose of morphine is 0.1 mg/kg and it can be given intravenous (IV), intramuscular (IM) or subcutaneous. It may be repeated after 5 minutes. The ventilation facilities should be at hand
- 87. The other alternative sedatives are: Midazolam 0.05–0.1 mg/kg (IV, intranasal or intrarectal) or Dexmedetomidine 0.5 -1 mcg/kg IV or Fentanyl 1–2 mcg/kg IV Ketamine has dual benefit of causing sedation and increasing SVR. The dose is 0.25- 1.0 mg/kg IV or IM.
- 88. 3. 100% Oxygen supplementation This causes pulmonary vasodilation and hence decreases the pulmonary vascular resistance (PVR)
- 89. 4. Sodium bicarbonate in a dose of 1–2 meq/kg IV is given slowly to correct metabolic acidosis This may reduce the respiratory centre stimulating effect of acidosis and may diminish the increase in pulmonary vascular resistance caused by hypoxia and acidosis It can be repeated in 10-15 minutes
- 90. 5. Beta blockers like injection propanolol is given in a dose of 0.1-0.2 mg/kg IV over 5 minutes and can be repeated once after 15 minutes It decreases the heart rate, infundibular spasm and increases SVR If propanolol is not available then injection metoprolol can be given
- 91. 6. In refractory cases vasopressors can be given to increase the SVR and promote the redirection of blood flow through the pulmonary circulation Phenylephrine a alpha-adrenergic blocker can be given in a dose of 5 to 20 mcg/kg IV bolus, followed by an infusion of 0.1 to 0.5 mcg/kg/min
- 92. 7. Avoid any actions that agitate the baby like vigorous examination, repeated attempts to venipuncture etc The drugs to be avoided are inotropes (e.g. digoxin, dopamine, or dobutamine) and diuretics.
- 93. If the spell is persistent or refractory, then intubation and mechanical ventilation maybe required A emergency Blalock-Taussig (BT) shunt / pulmonary balloon valvuloplasty (PBV) may be required in refractory cases
- 94. Transcatheter Interventions In TOF Balloon dilatation of pulmonary stenosis Balloon dilatation and/or ductal stenting Balloon dilatation of peripheral pulmonary artery stenosis with or without stenting Stenting of RVOT for infundibular stenosis by balloon expandable stainless steel stents (Johnson & Johnson). Transcatheter pulmonary valve replacement
- 95. Pulmonary Balloon Valvuloplasty Balloon dilatation of pulmonary stenosis may be an effective palliative procedure in a subset of patients, obviating the need for a palliative shunt The PBV is recommended if the patient’s size or cardiac anatomy makes that patient an unsuitable candidate for total surgical correction
- 96. The valvar obstruction should be a significant part of the RVOTO obstruction Also due to the multiple obstructions in the RVOT, the subvalvar obstruction still remains thus preventing flooding of the lungs after PBV The supravalvar pulmonic stenosis, if discrete, can be relieved by balloon dilatation With the balloon dilatation, immediate surgical intervention with high risk is avoided
- 98. Advantages of Pulmonary Balloon Valvuloplasty Substantial increase in saturation (SO2) Growth of pulmonary valve annulus and pulmonary arteries The need for transannular patch is reduced by 40 percent The high risk intracardiac repair (ICR) is postponed in infants PBV in TOF acts as a safe bridge to surgery.
- 99. Disadvantages of Pulmonary Balloon Valvuloplasty Pulmonary balloon valvuloplasty may not be successful in all patients Very rarely in severely hypoxic and sick patients with very low SO2 the very attempt to cross the infundibulum can precipitate the cyanotic spell The mortality can occur due to either cyanotic spell or tamponade in very sick infants
- 101. Shunt operations Designed to reduce cyanosis Pott shunt Waterston shunt Blalock-Taussig shunt
- 102. Blalock-Taussing shunt (classical) Subclavian artery to pulmonary artery anastomosis (end-to-side) Infrequently, this may lead to pulmonary hypertension
- 103. Blalock Taussing shunt (Classical) Originally, the shunt sacrificed the subclavian artery (with a distal ligation) and the proximal portion is routed downwards to an end to side anastomosis with the ipsilateral branch of the pulmonary artery
- 105. Blalock Taussing shunt (Modified) Uses a synthetic graft, usually expanded polytetrafluoroethylene (Gore-Tex), to connect the arteries Interposition graft between subclavian artery and ipsilateral pulmonary artery
- 107. Pott shunt It consists of a shunt formed between the descending thoracic aorta and the left pulmonary artery
- 108. Waterston shunt It consists of a shunt formed between the ascending aorta and the right pulmonary artery
- 109. Surgery The ideal age for TOF repair remains controversial Most centers prefer to operate by 1 year of age Transannular-transpulmonary approach is usually followed
- 110. Surgical Techniques for Intracardiac Repair
- 112. Indices for prediction of success of intracardiac repai
- 113. Nakata index Sum of the cross sectional areas of the left and right pulmonary arteries at their prebranching points as related to body surface area The normal Nakata index is + 330 mm²/m² An index of more than 150 mm²/m² is acceptable for complete repair without prior palliative shunt Tetralogy of Fallot with pulmonary stenosis should have an index of more than 100 for surgery
- 114. McGoon ratio Ratio of the sum of the pre branching diameters of the left and right pulmonary arteries to the diameter of the descending aorta just above the level of the diaphragm Ratio above 1.2 is associated with acceptable postoperative right ventricular systolic pressure in tetralogy of Fallot
- 119. Pulmonary atresia with Fallot’s tetralogy is the ultimate expression of severity The right ventricle terminates blindly against an atretic pulmonary valve or against imperforate muscle The pulmonary trunk is either a vestigial cord or a hypoplastic funnel-shaped channel that widens as it approaches the bifurcation
- 120. The proximal pulmonary arteries are hypoplastic and may be discontinuous The entire right ventricular output enters the systemic circulation via the nonrestrictive malaligned ventricular septal defect
- 122. The lungs are perfused by systemic-to-pulmonary arterial collaterals on which survival depends Exceptionally, the pulmonary circulation is supplied primarily, if not exclusively, by a long, narrow sigmoid-shaped ductus arteriosus
- 123. MAPCAs Systemic arterial collaterals are classified according to their origins as: Bronchial Originate where their name indicates and anastomose to pulmonary arteries within the lung Direct systemic arterial collaterals Originate from the descending aorta, enter the hilum, and then assume the structure and distribution of intrapulmonary arteries Indirect systemic arterial collaterals Originate from the internal mammary, innominate, and subclavian arteries and anastomose to proximal pulmonary arteries outside the lung
- 124. Systemic arterial collaterals anatomose with pulmonary arteries in three locations: (1) intrapulmonary; (2) extrapulmonary; and (3) hilar All three major types of collaterals are present when Fallot’s tetralogy occurs with pulmonary atresia, but only bronchial collaterals are present when the tetralogy occurs with pulmonary stenosis, irrespective of severity About 10% of arterial collaterals originate from coronary arteries
- 126. FALLOT’S TETRALOGY WITH ABSENT PULMONARY VALVE
- 127. Pulmonary valve tissue is lacking completely or consists of rudimentary remnants of avascular myxomatous connective tissue Rarely, absence of the pulmonary valve occurs with absence of a pulmonary artery and with systemic to pulmonary artery collaterals Obstruction to right ventricular outflow resides at the narrow pulmonary annulus, not at the malaligned infundibular septum The pulmonary trunk, especially its proximal branches, dilates massively together with the infundibulum
- 128. Management The life-threatening symptom is respiratory distress in the newborn It occurs predominantly in the Fallot-type APV Airway management as a primary procedure involves intubation, mechanical ventilation and extracorporeal membrane oxygenation in some infants Urgent complete surgical repair should relieve the compression of the tracheobronchial tree This is achieved by combined anterior and posterior plication of the pulmonary arteries or by translocation of the pulmonary artery anterior to the aorta and away from the airways, ‘maneuver de Lecompte’ procedure
- 129. Repair of pulmonary insufficiency and stenosis requires placement of a valve conduit (homograft or heterograft) in the right ventricular outflow tract Repair in the Fallot-type APV includes additional closure of the VSD with a patch Asymptomatic infants can undergo repair within the first 6 to 12 months Repair should however not be delayed for too long in order to avoid the harmful effect of the dilated pulmonary arteries on the tracheobronchial tree