Introduction to Transfection.pdf
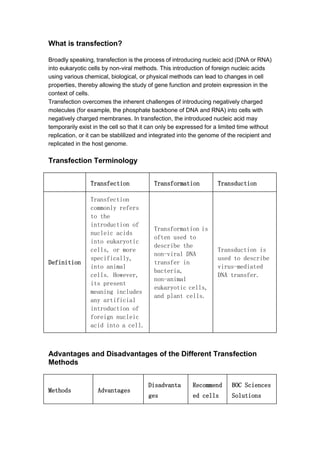
- 1. What is transfection? Broadly speaking, transfection is the process of introducing nucleic acid (DNA or RNA) into eukaryotic cells by non-viral methods. This introduction of foreign nucleic acids using various chemical, biological, or physical methods can lead to changes in cell properties, thereby allowing the study of gene function and protein expression in the context of cells. Transfection overcomes the inherent challenges of introducing negatively charged molecules (for example, the phosphate backbone of DNA and RNA) into cells with negatively charged membranes. In transfection, the introduced nucleic acid may temporarily exist in the cell so that it can only be expressed for a limited time without replication, or it can be stablilized and integrated into the genome of the recipient and replicated in the host genome. Transfection Terminology Transfection Transformation Transduction Definition Transfection commonly refers to the introduction of nucleic acids into eukaryotic cells, or more specifically, into animal cells. However, its present meaning includes any artificial introduction of foreign nucleic acid into a cell. Transformation is often used to describe the non-viral DNA transfer in bacteria, non-animal eukaryotic cells, and plant cells. Transduction is used to describe virus-mediated DNA transfer. Advantages and Disadvantages of the Different Transfection Methods Methods Advantages Disadvanta ges Recommend ed cells BOC Sciences Solutions
- 2. Chemical Lipid mediated High efficiency-able to effectively deliver nucleic acids into cells and obtain measurable changes Low cytotoxicity Easy to use-minimum steps required; adapt to high throughput systems Economi cal-more active lipids can reduce costs and obtain effective results Not suitable for all cell types-some cell lines cannot be transfected by lipofection Immortal cells, adherent (attached ) or suspended cells Most of our products are based on cationic lipid Calcium phosphate High-effi ciency Cheap Can be applied to multiple cell types Small pH changes (±0.1) may affect the efficiency The size and quality of the sediment are critical to success Adherent and nonadhere nt cell lines. Calcium Phosphate Transfection Reagent Cationic Polymers (e.g., PEI) No viral vector Chemi cal toxicity to some cell types Tumor cell lines PEI Transfection Reagent DEAE-Dextran Cheap Easy to perform and quick High concentrations of DEAE-dextran
- 3. Suitable to a wide range of cell types may be toxic to cells Differ ent transfection efficiency in different cell types Can only be used for transient transfection Poor performance in primary cells (usually < 10%) Magnet mediated Fast Improve transfection efficiency through targeted transport, especially for small amounts of nucleic acids Gentle treatment of cells Relativ ely new method Need adherent cells; suspension cells need to be fixed or centrifuged Immortal cells, Adherent mammalian cell lines and primary cells Biological Viral mediated Very high gene delivery efficiency, 95-100% Easy to use Effective for dissociated cells, slices, and in vivo Most viruses need P2 containment Risk of foreign gene integration into the host genome Many viruses are lytic Attached adherent cells, stem cells, primary cells Virus DNA Transfection Reagent Lentivira l Transfection Reagent Polybren e-Based Virus Delivery Reagent
- 4. Requir es packaging cell line Immunogenicit y DNA package size limit Physical Electroporat ion No need for vector Easy and rapid Not alter target cell morphology and functions Dema nds experimenter skill, laborious procedure Need to determine the optimum electroporatio n conditions Can cause cell death if transfection is not performed under optimum conditions A variety of cells (especial ly cells that are difficult to transfect , such as primary and stem cells) Electroporat ion Products Factors affecting transfection efficiency Cell passage number Keep the number of passages<50. In addition, the number of cell passages used in various experiments should be consistent. Cell state Cells should be grown in a medium suitable for the cell line and supplemented with serum or growth factors according to survival needs. Contaminated cells and media must not be used for transfection. If the cells are damaged in any way, they should be discarded and re-seeded from frozen, uncontaminated stock. Cell density
- 5. Normally, the confluency of the transfected cells is 40-80%. Too few cells will cause the culture to grow poorly without cell-to-cell contact. Too many cells can lead to contact inhibition, which prevents the cells from absorbing foreign DNA. Nucleic Acid Quality and Quantity The plasmid DNA used for transfection should be free of protein, RNA, chemical, and microbial contamination, and the appropriate final concentration is 0.2-1mg/ml. The optimal amount of DNA used for transfection depends on the type of DNA, transfection reagent, target cell line, and the number of cells. Cytotoxicity of transfection reagent When choosing a transfection method or reagent, both the transfection efficiency and the level of cytotoxicity must be considered. Higher transfection efficiency contributes to greater success. However, low toxicity methods cannot be ignored, because highly cytotoxic methods can cause adverse effects in the form of visible morphological changes and unknown changes in gene expression or stress response pathways. Transfection classification and application Transfection is used to study the function and regulation of genes or gene products, to produce genetically modified organisms, and is used as a powerful analysis tool for gene therapy methods. According to the time range of the desired experiment and the goal, transfection can be divided into transient transfection and stable transfection. Transient transfection Transiently transfected cells are usually harvested within 24-96 hours after transfection. They are usually used to study the short-term expression of genes or gene products, perform RNA interference (RNAi)-mediated gene silencing, or rapidly-produce recombinant proteins. Stable transfection In order to obtain long-term gene expression, the DNA vector needs to be integrated into the host chromosome. This process requires selective screening and clonal isolation. It is usually used for long-term pharmacological research, gene therapy, or long-term genetic regulation mechanism research.