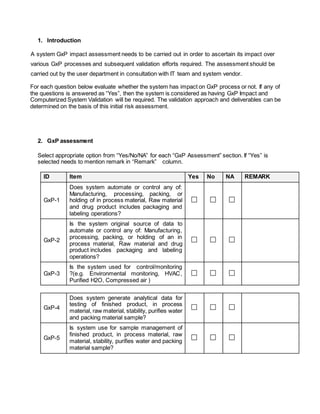
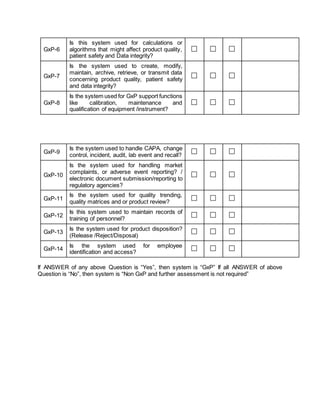
This document provides a framework for assessing whether a system has impact on Good Manufacturing Practices (GxP) processes and determining the appropriate validation approach. It involves evaluating 14 questions to identify if the system is considered GxP. If any question is answered yes, the system has a GxP impact. It also evaluates if the system stores or allows electronic signatures for electronic records required by regulators. Finally, it categorizes the software based on the GAMP 5 framework as an operating system, standard software package, configurable software package, or custom software. The assessment aims to establish the level of validation required.

![7 Does the system consist of a program or group of programs specially
designed for the end users?
4.2 Based on above mentioned GAMP 5 categorization checklist Application software falls
into one of the categories as:
GAMP 5
category
Checklist Evaluation
Category 1 Operating System
[Note: If the answer to question 1 to 3 is Yes, then category 1 is
applicable]
Category 3 Standard Software Packages (Commercial Off the Shelf (COTS) As-Is)
[Note: If the answer to question 4 and 5 is Yes, then category 3 is
applicable]
Category 4 Configurable Software Packages (Configurable COTS)
[Note: If the answer to question 4 and 6 is Yes, then category 4 is
applicable]
Category 5 Custom (Bespoke) Software
[Note: If the answer to question 7 is Yes, then category 5 is applicable.]](https://image.slidesharecdn.com/gxpassessment-220820080649-ae176423/85/GxP-Assessment-docx-4-320.jpg)