Synaptic Local Enrichment of CaMKIIα for Memory Improvement in Mice.pptx
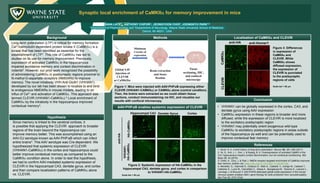
- 1. Synaptic local enrichment of CaMKIIa for memory improvement in mice Background Methods AAV-PHP.eB enables systemic expression of CLEVIR Localization of CaMKIIa and CLEVIR Conclusion SANA LATIF1,, ANTHONY CHIFOR2,, JEONGYOON CHOI2, JOONGKYU PARK1,2 1Department of Pharmacology and 3Department of Neurology, Wayne State University School of Medicine Detroit, MI 48201, USA Long-term potentiation (LTP) is critical for memory formation. Ca2+/calmodulin-dependent protein kinase II (CaMKIIa) is a kinase that has been identified as essential for the establishment of LTP1. This role of CaMKIIa has led to studies on its use for memory improvement. Previously, expression of activated CaMKIIa in the hippocampus impaired avoidance memory and context discrimination in rodents2. However, our prior work recognized the possibility of administering CaMKIIa in postsynaptic regions proximal to N-methyl-D-aspartate receptors (NMDARs) to improve memory. The novel intrabody VHH Anti-GluN1 (VHHAN1) developed by our lab has been shown to localize to and bind to endogenous NMDARs in mouse models, leading to an influx of Ca2+ and activation of CaMKIIa. This approach was termed CLEVIR (VHHAN1-CaMKIIa).3 Local enrichment of CaMKIIa by the intrabody in the hippocampus improved contextual memory3. References • VHHAN1 can be globally expressed in the cortex, CA3, and dentate gyrus using AAV expression • CaMKIIa expression in these regions is broader and more diffused, while the expression of CLEVIR is more localized to the excitatory postsynaptic region • VHHAN1 may potentially orient exogenous wild-type CaMKIIa to excitatory postsynaptic regions in areas outside of the hippocampus as well and can be potentially used to improve contextual fear memory 1. Nicoll, R. A. A brief history of long-term potentiation. Neuron 93, 281–290 (2017) 2. Ye, S., Kim, J. I., Kim, J. & Kaang, B. K. Overexpression of activated CaMKII in the CA1 hippocampus impairs context discrimination, but not contextual conditioning. Mol Brain 12, 32 (2019) 3. Chifor, A., Choi, J. & Park J. NMDA receptor-targeted enrichment of CaMKIIa improves fear memory. bioRxiv doi: https://doi.org/10.1101/2021.11.06.467164. 4. Morabito, G., Giannelli S., Ordazzo G., Bido S., Castoldi, V., Indrigo M., Cabassi T., Cattaneo S., Luoni M., Cancellieri C., Sessa A., Bacigaluppi M., Taverna S., Leocani L., Lanciego J. & Broccoli V. AAV-PHP.B-dedicated global-scale expression in the mouse nervous system enables GBA1 gene therapy for wide protection from synucleinopathy. Mol Ther 25: 2727-2742. HA-CaMKIIa VHHAN1- HA-CaMKIIa CaMKIIa CLEVIR Figure 2: Systemic expression of HA-CaMKIIa in the hippocampal CA3, dentate gyrus, and cortex in comparison to VHHAN1-HA-CaMKIIa Scale bar = 50 mm Hippocampal CA3 Cortex Dentate Gyrus anti-HA anti-HA anti-Homer1 Hypothesis Since memory is linked to the cerebral cortices, it is possible that applying the CLEVIR approach to broader regions of the brain beyond the hippocampus can improve memory better. This was accomplished using an AAV-DJ serotype known as AAV-PHP.eB which can infect entire brains4. This AAV serotype was Cre-dependent. We hypothesized that systemic expression of CLEVIR (VHHAN1-CaMKIIa) in the cortex and hippocampus could better improve contextual memory as compared to the CaMKIIa condition alone. In order to test this hypothesis, we had to confirm AAV-mediated systemic expression of CLEVIR in the hippocampal CA3, dentate gyrus, and cortex and then compare localization patterns of CaMKIIa alone vs. CLEVIR. Global AAV injection of CLEVIR or CaMKIIa Minimum 3 weeks of expression Brain extraction and tissue fixation Tissue sectioning, IHC, and confocal microscopy Figure 1: Mice were injected with AAV-PHP.eB expressing either CLEVIR (VHHAN1-CAMKIIa) or CAMKIIa alone (control condition). Then, the brains were extracted so we could obtain tissue sections, conduct immunostaining via IHC, and visualize our results with confocal microscopy. Figure 3: Differences in expression of CaMKIIa and CLEVIR. While CaMKIIa shows diffused expression, the expression of CLEVIR is punctated to the postsynaptic regions of cells Scale bar = 50 mm
