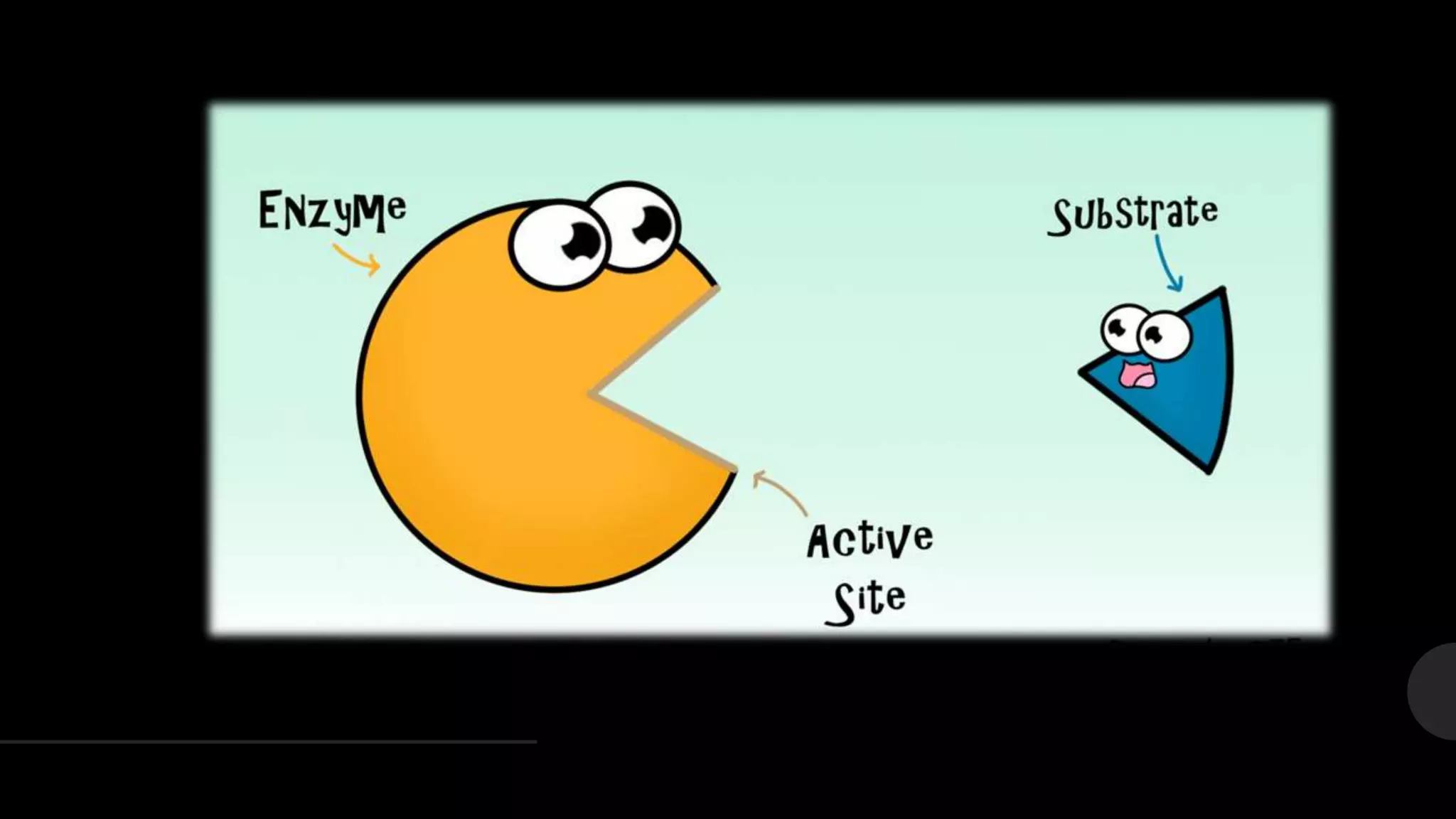
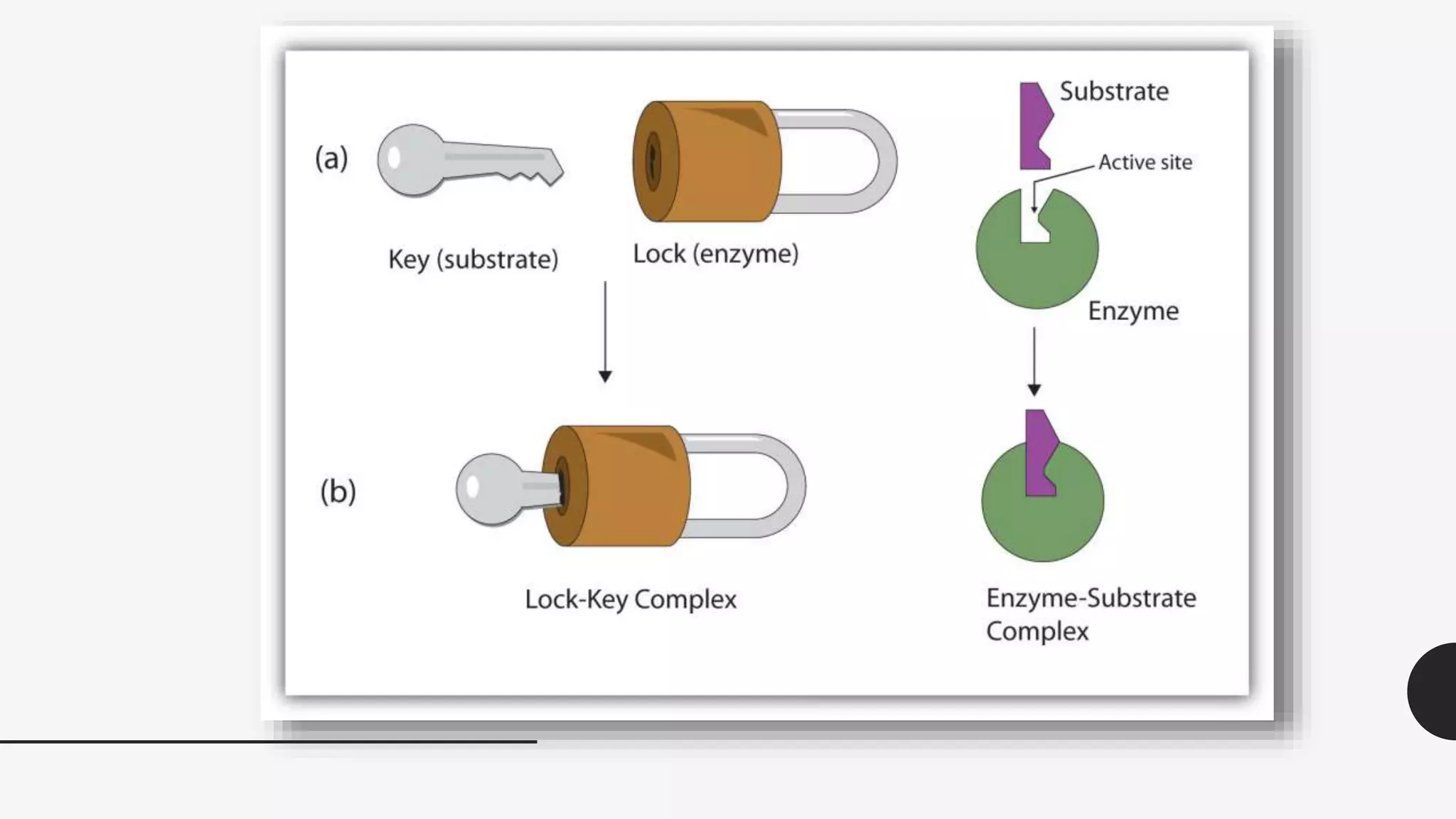
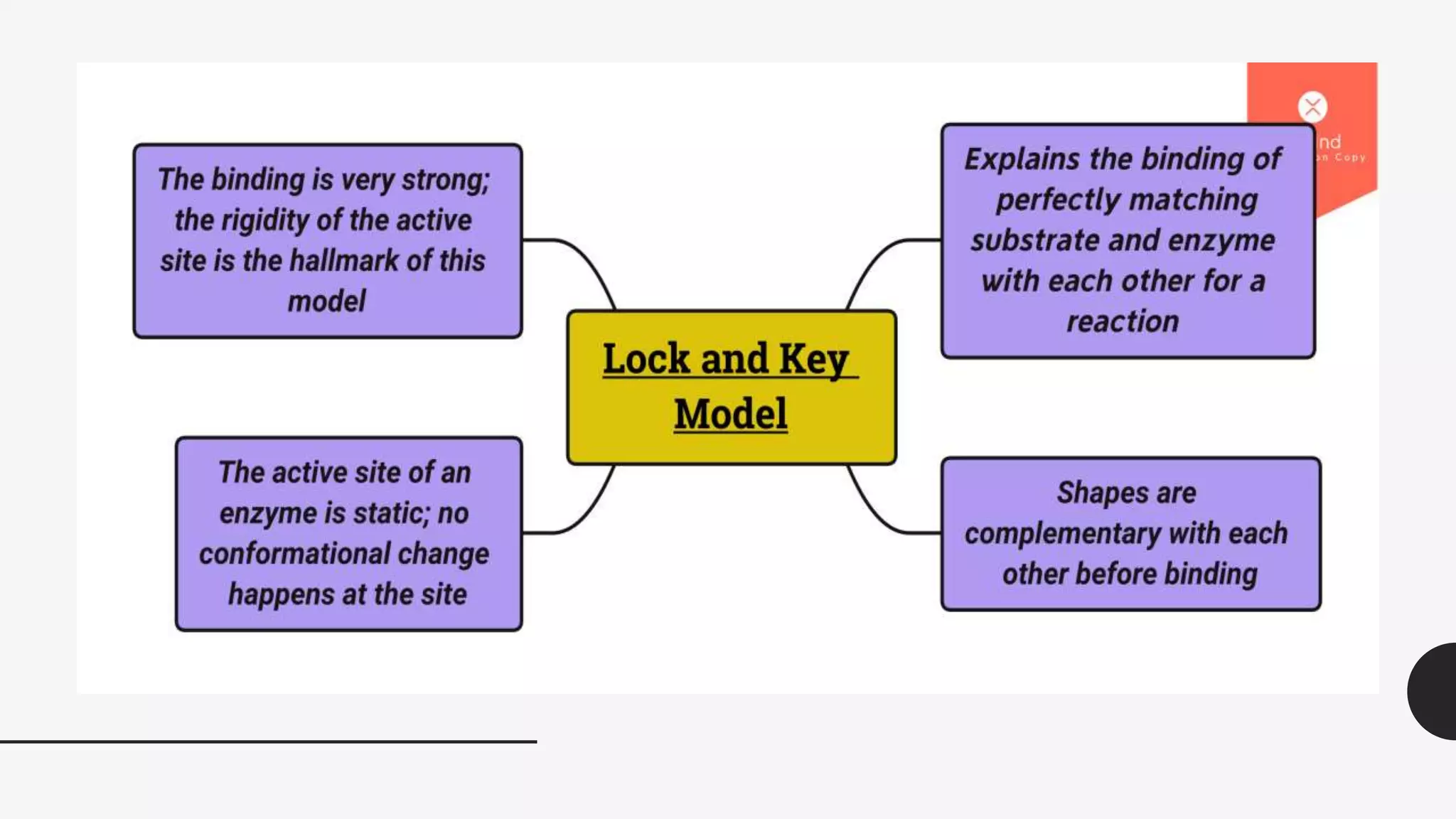
The document summarizes the lock-and-key hypothesis proposed by Emil Fischer in 1894 and the induced fit hypothesis proposed by Daniel Koshland in 1958 to describe enzyme-substrate binding. The lock-and-key hypothesis suggests that enzymes and substrates have complementary geometric shapes that fit exactly. The induced fit hypothesis refined this by proposing that the enzyme's active site is flexible and can change shape upon substrate binding to optimize the interaction. Both hypotheses helped explain the specificity of enzymes for their substrates.