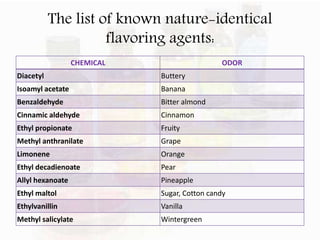
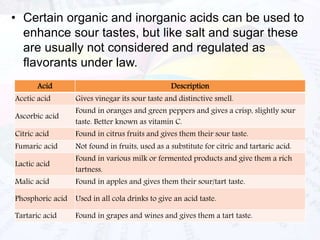
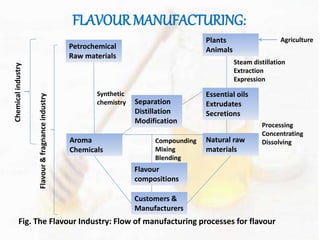
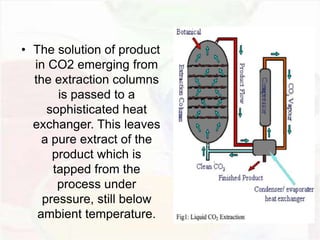
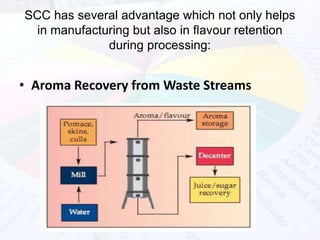
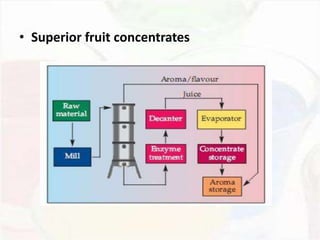
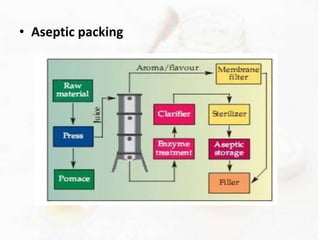
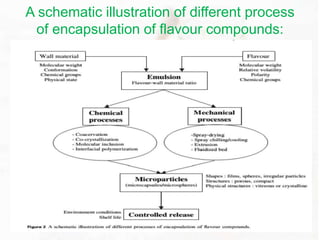
This document provides an overview of flavours used in food industries. It discusses the history and classification of flavours, as well as methods for producing natural flavours through extraction, distillation, and biotechnology. It also covers techniques for producing artificial flavours synthetically. The document aims to educate readers on the various flavour manufacturing processes and categories used in food production.