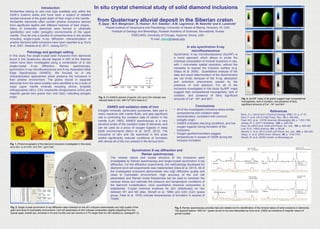
Agu poster 1
- 1. In situ crystal chemical study of solid diamond inclusions from Quaternary alluvial deposit in the Siberian craton P. Dera1* , M.H. Manghnani1 , A. Hushur1 , N.V. Sobolev2 , A.M. Logvinova2 , M. Newville3 and A. Lanzirotti3 1 Hawaii Institute of Geophysics and Planetology, University of Hawaii at Manoa, Honolulu, HI, USA 2 Institute of Geology and Mineralogy, Russian Academy of Sciences, Novosibirsk, Russia 3 GSECARS, University of Chicago, Argonne, Illinois, USA *E-mail: pdera@hawaii.edu Introduction Kimberlites belong to rare rock type available only within the Earth’s cratonic areas and have been a subject of detailed studies because of the great depth of their origin in the mantle. Kimberlitic diamonds often contain pristine inclusions derived from significant depths with different histories of their origins. Many of kimberlitic diamonds were formed in ultramafic (peridotitic) and mafic (eclogitic) environments of the upper mantle. Thus far only a handful of comprehensive in situ studies including single-crystal X-ray diffraction characterization of pristine diamond solid inclusions have been reported (e.g. Kunz et al. 2001, Nestola et al. 2011, Joswig 2011). In situ synchrotron X-ray microfluorescence Synchrotron X-ray microfluorescence (SµXRF) is a novel approach which allows to probe the chemical composition of mineral inclusions in situ, with 1 micrometer spatial resolution, without the necessity to expose the inclusion surface (e.g. Sitepu et al. 2005). Quantitative analysis of the data and exact determination of the stoichiometry are not trivial, because of the X-ray absorption and extinction phenomena caused by the presence of host diamond. For all of the inclusions investigated in this study SµXRF maps suggest high compositional homogeneity, lack of zonation, and presence of fairly significant amounts of Ca2+ , Ni2+ and Mn2+ . Synchrotron X-ray diffraction and Raman spectroscopy The mineral nature and crystal structure of the inclusions were investigated by Raman spectroscopy and single-crystal synchrotron X-ray diffraction. For the diffraction experiments, the methodology developed for diamond anvil cell experiments was implemented (Dera et al. 2013). All of the investigated inclusions demonstrate very high diffraction quality and close to hydrostatic environment. High accuracy of the unit cell parameters and Raman mode frequencies can be used to constrain the residual stress and estimate the pressure and temperature conditions of the diamond crystallization, once quantitative chemical composition is established. Crystal chemical evidence for oEn (distribution on iron between M1 and M2 sites, Stimpfl et al. 1999) and cOm (C2/c space group, Fleet et al. 1978) indicate temperatures of formation in excess of 1000K. Petrology and geologic setting In this study five single-crystal solid inclusions from diamonds found in the Quaternary alluvial deposit in NW of the Siberian craton have been investigated using a combination of in situ single-crystal X-ray diffraction, Raman spectroscopy, synchrotron X-ray microfluorescence and X-ray Absorption Near Edge Spectroscopy (XANES). We focused on in situ characterization approaches which preserve the inclusions in their pristine environment. The grains (found in separate diamonds from the same locality) were identified to be a suite of major upper mantle minerals including olivine, enstatite orthopyroxene (oEn), C2/c omphacite clinopyroxene (cOm) and majoritic garnet (two grains Ga1 and Ga2), indicating eclogitic origin. XANES and oxidation state of iron Eclogitic minerals, particularly pyroxenes, take part in redox reactions with mantle fluids, and play significant role in controlling the oxidation sate of carbon in the mantle (Luth 1993). XANES spectroscopy is a very sensitive probe of the oxidation state of elements, and can be used as a proxy of oxygen fugacity in deep Earth environments (Berry et al. 2010, 2013). The inclusions of oEn and Ga examined in this study exhibit significantly reduced conditions of formation with almost all of the iron present in the ferrous form. References Berry, A.J. (2010) et al. Chemical Geology, 278(1–2): p. 31-37. Dera, P. et al. (2013) High Press. Res., 33: p. 466-484. Fleet, M.E. et al. (1978) American Mineralogist, 63: p. 1100-1106. Joswig, W. Z. (2011) Kristallogr., 226: p. 226-228. Kunz, M. (2002) et al. Earth and Planet. Sci. Lett., 198: p. 485-493. Luth, R.W. (1993) Science, 261: p. 66-68. Nestola, F. et al. (2011) Earth and Planet. Sci. Lett., 305: p. 249-255. Sitepu, H. et al. (2005) Amer. Mineral., 90: p. 1740-1747. Stimpfl, M. et al. (2005) Contrib. to Mineralogy an Fig. 1. Photomicrographs of the diamond inclusions investigated in this study (a,b) oEn, (c,d) cOm, (e,f) Ga1, (g,h) Ga2 Fig. 2. Single crystal synchrotron X-ray diffraction data collected on the oEn inclusion demonstrate very high quality of the grain and close to hydrostatic environment. Unit cell parameters of oEn indicate composition slightly departing from the typical upper mantle opx, enriched in Fe and Ca (the unit cell volume is 0.7% larger than for oEn studied by Joswig(2011)). Fig. 5. Raman spectroscopy provides fast and reliable tool for identification of the mineral nature of solid inclusions in diamonds. The spectral feature ~900 cm-1 (green arrow) in Ga was interpreted by Kunz et al. (2002) as indicative of majoritic nature of garnet crystals. Fig. 3. Fe XANES spectra of garnet, oEn and cOm indicate very reduced state or iron, with Fe3+ /ΣFe close to 0. Fig. 4. SµXRF maps of all grains suggest high compositional homogeneity, lack of zonation, and presence of fairly significant amounts of Ca2+ , Ni2+ and Mn2+ . Conclusions • All of the investigated inclusions share similar compositional and oxidation state characteristics, consistent with common eclogitic origin. • XANES indicates reducing conditions, and low oxygen fugacity during formation of the inclusions. • Oxygen geothermometers suggest temperatures in excess of 1000K during the inclusion formation. a) b) c) d) e) f) g) h)