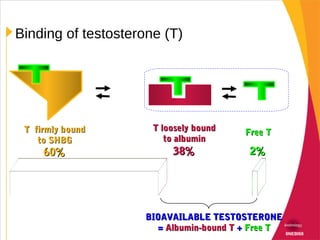
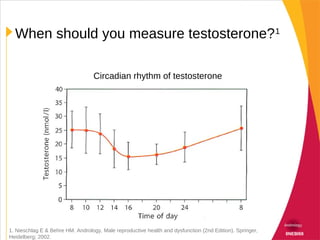
This document discusses hypogonadism and testosterone replacement. It provides information on:
- The causes and clinical presentation of primary and secondary hypogonadism.
- Diagnosing hypogonadism through patient history, physical exam, and measuring serum testosterone and other hormone levels.
- Goals of testosterone replacement therapy in treating symptoms and restoring physiological functions.
- Various treatment options for testosterone replacement therapy including oral, buccal, implant, patch, gel, and intramuscular injection formulations. It provides details on the administration and pharmacokinetics of these different options.