Chemical bond 2.ppt
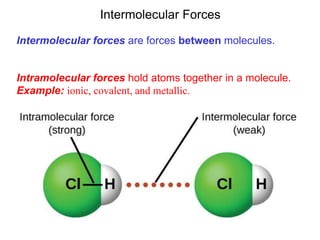
- 1. Intermolecular Forces Intermolecular forces are forces between molecules. Intramolecular forces hold atoms together in a molecule. Example: ionic, covalent, and metallic.
- 2. Electronegativity is the ability of an atom to attract toward itself the electrons in a chemical bond. Electronegativity - relative, F is highest H F electron poor region electron rich region
- 3. H F d+ d-
- 4. e- e- δ - δ + 2 + e- e- 2 + e- e- 2 + e- e- δ - δ + 2 + e- e- δ - δ + 2 + e- e- δ - δ + 2 + repel attract e- e- 2 +
- 5. Types of Intermolecular Forces Dipole-Dipole Forces Attractive forces between polar molecules Orientation of Polar Molecules in a Solid
- 6. Dipole – Dipole Forces d+ d- d+ d- d+ d- d+ d- d+ d-
- 7. Types of Intermolecular Forces Ion-Dipole Forces Attractive forces between an ion and a polar molecule Ion-Dipole Interaction
- 8. Ion – Dipole Forces Exist when we dissolve an ionic compound in a polar solvent, like H2O. The d- end of H2O is attracted to a cation (+). The d+ end of H2O is attracted to an anion (-).
- 9. Types of Intermolecular Forces Dispersion Forces – van der Walls forces/London forces (weakest) Attractive forces that arise as a result of temporary dipoles induced in atoms or molecules ion-induced dipole interaction dipole-induced dipole interaction
- 10. Hydrogen Bonding Hydrogen Bond = an attraction between a slightly positive hydrogen atom and a slightly negative atom, often oxygen or nitrogen
- 11. Hydrogen Bonding
- 12. O H H δ + δ + δ- O H H δ + δ + δ- O H H δ + δ + δ- O H H δ + δ + δ- O H H δ δ + δ- hydrogen bonds
- 13. • A hydrogen bond is an intermolecular bond. F—H O—H N—H • A hydrogen bond is formed between polar molecules that contain hydrogen covalently bonded to a small, highly electronegative atom: F, O, N.
- 14. hydrogen bond covalent bond covalent bond A dipole-dipole bond bond will be formed between the two molecules which is called a hydrogen bond.
- 15. Remember, you need: A hydrogen atom covalently bonded to an electronegative atom … N, O or F. If only one of these conditions is met, you don’t get hydrogen bonding. A lone pair of electrons on the electronegative atom.
- 16. Coordinate Covalent Bond • A coordinate bond (also called a dative covalent bond) is a covalent bond (a shared pair of electrons) in which both electrons come from the same atom. • A coordinate covalent bond is usually shown with an arrow.
- 17. Amino-Borane N has a full octet an a lone pair of electrons. NH3 is an uncharged molecule. B only has 6 electrons, it is deficient. BH3 is an uncharged molecule. N can “donate” its lone pair to B forming a co-ordinate bond.
- 18. Amino-Borane Now we have a new molecule with a covalent bond between N and B. While there are formal charges on N and B, the overall charge on the molecule is neutral. The bond formed between N and B is just as strong as any covalent bond.
- 19. ammonium ion