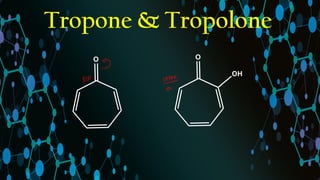
Tropone & Tropolones Aromaticity
- 2. Aromaticity is a Suspicious Concept CSE 2021 ❖ Aromatic Hydrocarbon with 16 π Electron Periphery
- 3. Tropone, Tropolone & Cycloheptanone Tropone Tropolone Cycloheptanone Hydrogenation Readily hydrogenated over Pd Not hydrogenated over Pd -- C=O stretching frequency (cm-1) 1638 1615 1699 Dipole Moment (D) 4.17 3.71 3.04 Resonance Energy (Kcal/mol) 29 36 -- Stability Less stable More stable -- Aromaticity Weakly aromatic Marked aromatic character (Reason: Intramolecular H- bonding + tautomerism) --
- 4. Aromaticity of Tropone ❖ Tropone display limited aromatic character ❖ Tropone is a resonance hybrid of the classical structure and tropylium cation structure ❖ React like Triene: readily hydrogenated, rapidly decolorizes aqueous permanganate ❖ Ionic structure contribution: high Dipole moment (4.17 D) indicating contribution from the polarized form, C=O stretching frequency (1638 cm-1) ❖ Classical structure contribution: Shows bond length alteration ❖ Basicity: more basic than normal ketone
- 5. Aromaticity of Tropolone ❖ Tropolone nucleus is not reduced by hydrogen in the presence of Pd catalysts but Raney nickel or Adams’ platinum oxide used as catalysts ❖ Both H-bonding and resonance with the ionic form (C) help to increase the stability of tropolone compared not only to tropone but also to the isomeric hydroxytropones ❖ In isomeric hydroxytropones: Intramolecular H-bonding must be absent and corresponding ionic forms involve greater separation of charge
- 6. Reaction with Electrophiles CSE 2012
- 7. Tropolone both fairly acidic and basic ❖ It is acidic because capable of loosing a proton to form relative stable anion while it is basic because of its ability to receive a proton to form cation. ❖ Intramolecular H-bonding is significantly diminished in 3 comparison to 1
- 8. ❖ Tropolone can be regarded as extended or vinylogous carboxylic acid and enol form of 1,3-diketones. ❖ on the simple basis of vinylogy, acidity of tropolone falls slightly below expectation ❖ acidity is certainly not enhanced by aromatic resonance of the ion (a). ❖ Resonating ions of the comparison substances are likewise fully equivalent and any additional “aromatic” resonance would have to come from structures (b) which must be rather less favored than the corresponding structures (c) of the free acid. ❖ Basicity: This property is a direct result of the aromaticity, In this sense it contrasts with the acidity of tropolones, which is not aided by that aromaticity. a b c Is Tropolone stable in acidic or basic medium?
- 9. IR Spectrum ❖ emphasis on the carbonyl and hydroxyl bands ❖ carbonyl group: exceptionally low wave number ❖ Reasons: Ring Size, High degree of conjugation, High polarity, Hydrogen-Bonding Effects ❖ Ring Size: IFoS 2006
- 10. IR Spectrum ❖ Conjugation effect: Increase single bond character od C=O, hence lowers force constant ❖ Hydrogen-Bonding Effects: Hydrogen bonding to a carbonyl group lengthens the C=O bond and lowers the stretching force constant K, resulting in a lowering of the absorption frequency. ❖ 1,3-Diketones:The carbonyl group in the enol form appearing at about 1622 cm−1 is substantially shifted and intensified in comparison to the normal ketone value, 1715 cm−1. The shift is a result of internal hydrogen bonding
- 11. Tropone Umpolung ❖ Conversion of the carbonyl group into a hydrazone ion reverses the polarity of the exocyclic double bond, increases the [4n] ring π- antiaromaticity. IFoS 2006 Diazocycloheptatriene: antiaromaticity effects in this derivative are completely attenuated.
- 12. Synthesis & Reaction ❖ Synthesis of tropone ❖ Synthesis of tropolone ❖ Rearrangement to benzoic acid