structure of atom
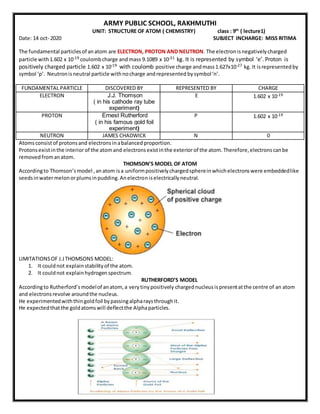
- 1. ARMY PUBLIC SCHOOL, RAKHMUTHI UNIT: STRUCTURE OF ATOM ( CHEMISTRY) class : 9th ( lecture1) Date: 14 oct- 2020 SUBJECT INCHARGE: MISS RITIMA The fundamental particlesof anatom are ELECTRON, PROTON AND NEUTRON. The electronisnegativelycharged particle with1.602 x 10-19 coulombcharge andmass 9.1089 x 10-31 kg. It is represented by symbol ‘e’. Proton is positively charged particle 1.602 x 10-19 with coulomb positive charge andmass1.627x10-27 kg. It isrepresentedby symbol ‘p’. Neutronisneutral particle withnocharge andrepresented bysymbol‘n’. FUNDAMENTAL PARTICLE DISCOVERED BY REPRESENTED BY CHARGE ELECTRON J.J. Thomson ( in his cathode ray tube experiment) E 1.602 x 10-19 PROTON Ernest Rutherford ( in his famous gold foil experiment) P 1.602 x 10-19 NEUTRON JAMES CHADWICK N 0 Atomsconsistof protonsand electronsinabalancedproportion. Protonsexistinthe interior of the atomand electronsexistinthe exterior of the atom.Therefore,electronscanbe removedfromanatom. THOMSON’S MODEL OF ATOM Accordingto Thomson’s model ,anatom isa uniformpositivelychargedsphereinwhichelectronswere embeddedlike seedsinwatermelonorplumsinpudding.Anelectroniselectricallyneutral. LIMITATIONSOF J.JTHOMSONS MODEL: 1. It couldnot explainstabilityof the atom. 2. It couldnot explainhydrogenspectrum. RUTHERFORD’S MODEL Accordingto Rutherford’smodelof anatom,a verytinypositively chargednucleusispresentatthe centre of an atom and electronsrevolve aroundthe nucleus. He experimentedwiththingoldfoil bypassingalpharaysthroughit. He expectedthatthe goldatomswill deflectthe Alphaparticles.
- 2. Observations Inferences Alpha particles which had high speed moved straight through the gold foil Atom contains a lot of empty space Some particles got diverted a by slide angles Positive charges in the atom are not occupying much of its space Only one out of 12000 particles bounced back The positive charges are concentrated over a particular area of the atom. LIMITATIONS OF RUTHERFORD’S MODEL: 1. It could not explain stability of the atom. 2. It couldnot explainhydrogenspectrum. BOHR’S MODEL OF ATOM The main pointsof Bohr’s model of atomare: 1. An atomhas three typesof particlescalledasfundamentalparticles.These are electrons,Protonsandneutrons. 2. The protonsand neutronsare presentinthe centre.The electronsare presentaroundthe nucleus. 3. An electroniselectricallyneutral asthe numberof protonsisequal tothe numberof electrons. 4. The electronsare revolvingaroundthe nucleusinfixedpathwhichare calledas ENERGYLEVELS OR ENERGY SHELLS OR ORBITS. The shellsare countedintwoways 1 , 2 , 3 , 4 , 5 , 6 or k , L , M , N , O , P. The countingis fromcentre to outwards. 5. The variousenergylevelsare arrangedin orderof increasingenergy. The orderof energyis: 1 < 2 < 3 < 4 < 5 < 6 or k < L < M < M < N < O < P 6. The energyof electroninan atom is quantized. 7. There isno change in the energyof the electronsaslong as they keeponrevolvinginthe same energylevel. 8. The angular momentumof anelectroninan atomis quantized. 9. The change inenergytakesplace whenanelectronjumps fromone energy level toanother. If electronsgainenergyfromoutside itjumpsfromlowerenergylevel tothe higherenergylevel. If electron jumpsfromhigherenergylevel tolowerenergy level, itlosesenergy.
- 3. READ AND REREAD: 1. Name the sub- atomicparticlesof an atom. Ans: Electron, Protonand Neutron. 2. On the basesof Rutherfordmodel whichsubatomicparticle ispresentinthe modecule of anatom? Ans: Proton 3. State three waysinwhichprotonsdifferfrom electron. PROTONS ELECTROS It has one unitof positive charge. It has one unitof negative charge.I It has massof 1.6 x 10-24 It has massof 1.6 x 10-28 It ispresentinthe nucleusof atom. It ispresentinthe extranuclearpartof an atom. 4. The fundamental particlespresentinatomwhichare equal innumberare: a. Protonand electron b. Neutronand electron c. Protonand neutron d. Protonsand positrons 5. A protonisidentical with: a. An atomof hydrogen b. A molecule of hydrogen c. The nucleusof helium d. The nucleusof hydrogenatom
