Ubiquinone (Coenzyme Q, or Q) Electron Carrier
•Download as PPT, PDF•
1 like•404 views
Ubiquinone (Coenzyme Q, or Q)
Report
Share
Report
Share
Recommended
Purine & pyrimidine metabolism and disorders

Purine & pyrimidine metabolism and disordersInternational Medicine School - Management and Science University
More Related Content
What's hot
Purine & pyrimidine metabolism and disorders

Purine & pyrimidine metabolism and disordersInternational Medicine School - Management and Science University
What's hot (20)
Chapter 15 - principle of metabolic regulation - Biochemistry

Chapter 15 - principle of metabolic regulation - Biochemistry
[Brief]Structure and functions of hemoglobin and myglobin (Bio-Inorganic chem...![[Brief]Structure and functions of hemoglobin and myglobin (Bio-Inorganic chem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![[Brief]Structure and functions of hemoglobin and myglobin (Bio-Inorganic chem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
[Brief]Structure and functions of hemoglobin and myglobin (Bio-Inorganic chem...
Biosynthesis and degradation of porphyrin and heme

Biosynthesis and degradation of porphyrin and heme
Biochemistry ii protein (metabolism of amino acids) (new edition)

Biochemistry ii protein (metabolism of amino acids) (new edition)
Mechanism of action of Chymotrypsin & Lysozyme.pptx

Mechanism of action of Chymotrypsin & Lysozyme.pptx
Fatty acid oxidation ( Beta , Alpha omega and peroxisomal)

Fatty acid oxidation ( Beta , Alpha omega and peroxisomal)
Similar to Ubiquinone (Coenzyme Q, or Q) Electron Carrier
Similar to Ubiquinone (Coenzyme Q, or Q) Electron Carrier (20)
Electron Transport Chain & Oxidative Phosphorylation

Electron Transport Chain & Oxidative Phosphorylation
Electron transport chain( Oxidative phosphorylation)

Electron transport chain( Oxidative phosphorylation)
Describe the major components of the electron transport chain. How w.pdf

Describe the major components of the electron transport chain. How w.pdf
Electron transport chain and Oxidative phosphorylation

Electron transport chain and Oxidative phosphorylation
More from Hamid Ur-Rahman
More from Hamid Ur-Rahman (20)
Pentose Phosphate Pathway (Hexose Monophosphate Shunt)

Pentose Phosphate Pathway (Hexose Monophosphate Shunt)
Hormonal Regulation: glycolysis/glucogenesis-Glucose homeostasis

Hormonal Regulation: glycolysis/glucogenesis-Glucose homeostasis
Recently uploaded
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Recently uploaded (20)
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...

BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...

Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...
Measures of Central Tendency: Mean, Median and Mode

Measures of Central Tendency: Mean, Median and Mode
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Ubiquinone (Coenzyme Q, or Q) Electron Carrier
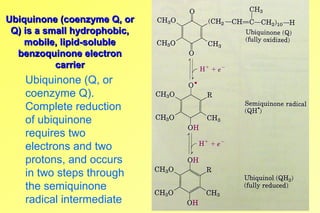
- 1. Ubiquinone (coenzyme Q, orUbiquinone (coenzyme Q, or Q) is a small hydrophobic,Q) is a small hydrophobic, mobile, lipid-solublemobile, lipid-soluble benzoquinone electronbenzoquinone electron carriercarrier Ubiquinone (Q, or coenzyme Q). Complete reduction of ubiquinone requires two electrons and two protons, and occurs in two steps through the semiquinone radical intermediate
- 2. Prosthetic groups of cytochromes. Each consists of four five-membered, nitrogen-containing rings in a cyclic structure called a porphyrin. The four nitrogen atoms are coordinated with a central Fe ion, either Fe2+ or Fe3+ . Iron protoporphyrin IX is found in b-type cytochromes and in hemoglobin and myoglobin (see Fig. 6-17). Heme C is covalently bound to the protein of cytochrome c through thioether bonds to two Cys residues. Heme A, found in the a-type cytochromes, has a long isoprenoid tail attached to one of the five-membered rings. The conjugated double-bond system (shaded in red) of the porphyrin ring accounts for the absorption of visible light by these hemes.
- 3. Iron-sulfur centers. The Fe-S centers of iron-sulfur proteins may be as simple as (a), with a single Fe ion surrounded by the S atoms of four Cys residues. Other centers include both inorganic and Cys S atoms, as in 2Fe-2S (b) or 4Fe-4S (c) centers. (Note that only the inorganic S atoms are counted in these designation. For example, in the 2Fe-2S center (b), each Fe ion is actually surrounded by four S atoms.) The exact standard reduction potential of the iron in these centers depends on the type of center and its interaction with the associated protein.
- 4. Summary of the flow of electrons and protons through the four complexes of the respiratory chain. Electrons reach Q through I and II. QH2 serves as a mobile carrier of electrons and protons. It passes electrons to Complex III, which passes them to another mobile connecting link, cytochrome c. Complex IV then transfers electrons from reduced cytochrome c to O2. Electron flow through Complexes I, III and IV is accompanied by proton flow from the matrix to the intermembrane space. Recall that electrons from β oxidation of fatty acids can also enter the respiratory chain through Q.
