Atkins Chapter1.ppt
- 1. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
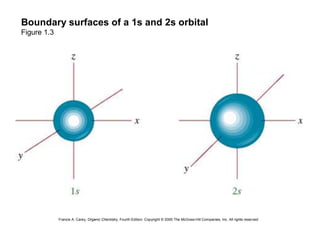
Boundary surfaces of a 1s and 2s orbital
Figure 1.3
- 2. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Boundary surfaces of the 2p orbitals
Figure 1.4
- 3. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
- 4. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Ionic bond
Figure 1.5
- 5. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
- 6. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
- 7. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
- 8. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
- 9. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Molecular models of methane
Figure 1.7
- 10. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
A wedge-and-dash drawing of the structure
of methane
Figure 1.8
- 11. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Shape of a molecule in terms of its atoms
Figure 1.9
- 12. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Representations of the trigonal planar geometry
of boron trifluoride
Figure 1.10
- 13. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Models of formaldehyde showing the trigonal planar
geometry of the bonds to carbon
Figure 1.11
- 14. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Linear geometry of carbon dioxide
Figure 1.12
- 15. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Contribution of individual bond dipole moments to
molecular dipole moments
Figure 1.13
- 16. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
sp3-Hybridization of carbon
Figure 1.20
- 17. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Orbital overlaps involved in bonds of methane
Figure 1.22
- 18. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Orbital overlap description
Figure 1.23
- 19. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Atoms of ethylene
Figure 1.24
- 20. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
sp2-Hybridization of carbon
Figure 1.25
- 21. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Orbital overlaps and
bonding in ethylene
Figure 1.27
- 22. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Acetylene is a linear molecule
Figure 1.28
- 23. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
sp-Hybridization of carbon
Figure 1.29
- 24. Francis A. Carey, Organic Chemistry, Fourth Edition. Copyright © 2000 The McGraw-Hill Companies, Inc. All rights reserved.
Orbital overlaps and bonding in acetylene
Figure 1.31