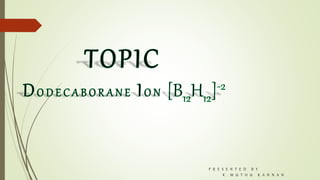
Dodecaborane anion
- 1. P R E S E N T E D B Y K . M U T H U K A N N A N
- 2. DODECABORANE: Introduction: The dodecaborate anion(12), [B12H12]2-, has the structure of a regular icosahedron of boron atoms, with each boron atom being attached to a hydrogen atom .The existence of the dodecaborate anion, [B12H12]2-,was predicted by H. C. Longuet-Higgins and M.de V. Roberts in 1955 on the basis of MO and LCAO calculations. This prediction was verified experimentally by Hawthorne and Pitochelli in 1960. It is also known as dodecahydro-closo- dodecaborate anion.
- 4. Preparations: It may be synthesised by the pyrolysis of the [B3H8]- ion . Diborane and borohydride reacts to give dodecaborane anion. One of the most convenient methods of synthesis of the closo-dodecaborate anion is pyrolysis of triethylamine-borane with decaborane(14) at 190 °C giving the desired product in 92% yield. B10H14 + 2 Et3N·BH3 → (Et3NH)2[B12H12] + 3 H2 5[B3H8]- [B12H12]-2 + 3[BH4]- + 8H2 2[BH4]- + 5B2H6 [B12H12]-2 + 13H2
- 5. Another convenient and widely used in laboratory practice method of synthesis the [B12H12]2-anion is the reaction of sodium tetrahydroborate with decaborane at 160 °C giving the product in 91% yield. B10H14 + 2 NaBH4 → Na2[B12H12] + 5 H2 Metallo derivatives: The reactions of the closo-dodecaborate anion with mercury acetate and trifluoroacetate gave metallated derivatives [B12H12–n(HgOC(O)CX3)n]2– (X = H, F; n = 1–12). However, in contrast to the mercurated carborane derivatives, the metallated dodecaborate derivatives are unstable and easily decompose in solution and in the solid state.
- 6. Derivative with boron-carbon bond: The reaction of the [B12H12]2– with carbon monoxide at 1000 atm and 130 °C in the presence of [Co2(CO)8] as a catalyst gives almost a quantitative conversion to a mixture of mono- and disubstituted carbonyl derivatives, [B12H11CO], [1,12B12H10(CO)2], and [1,7-B12H10(CO)2]. [B12H12]2– + CO → [B12H11CO]+ [1,7-B12H10(CO)2]+[1,12B12H10(CO)2]. Derivative with boron-nitrogen bond: The reaction of the closo-dodecaborate anion with hydroxylamine-O-sulfonic acid in aqueous solution giving mono- and disubstituted amino derivatives which, because of their high basicity, are readily isolated in the N-protonated forms.
- 7. The diamino derivative was obtained as a mixture of the 1,7-, 1,12-, and a small amount of 1,2-isomers. [B12H12]2– → [B12H11NH3]- → [1,2-, 1,7- and 1,12-B12H10(NH3)2] Derivative with boron-phosphate and boron-arsenic bond: The trimethylphosphonium derivatives of the dodecaborate anion were obtained by pyrolysis of the trimethylphosphine–borane complex, where the [B12H11PMe3]– yield reaches 60%. Me3P·BH3 → [B12H11PMe3]– + [B12H10(PMe3)2] The [1,7-B12H10(PMe2Ph)2] complex was obtained by the reaction of the closo-dodecaborate anion with [PdCl2(Me2PhP)2] in tetrahydrofuran.
- 8. [B12H12]2– + [PdCl2(Me2PhP)2] → [1,7-B12H10(PMe2Ph)2] The trimethylarsonium derivatives [B12H11AsMe3]– and [B12H10(AsMe3)2] were obtained by pyrolysis of the trimethylarsine– borane complex. Derivative with boron-oxygen bond: Alkylation of [B12H11OH]2– with alkyl halides in dimethyl sulfoxide in the presence of potassium hydroxide or in acetone in the presence of potassium carbonate gives the alkoxy derivatives [B12H11OR]2–. [B12H12OH]2- + RX → [B12H12OR]2- R = C2H5, (CH3)2CH, C6H5CH2
- 9. Derivative with boron-Sulphur bond: [B12H12]2– anion with thiourea under conditions of chemical oxidation. [B12H12] 2– + SC(NH2)2 → [B12H11SC(NH2)2]– + OH– → [B12H11SH]2– Derivative with boron-selenium bond: The selenocyanato derivative [B12H11SeCN]2– was prepared by the reaction of the closo-dodecaborate anion with (SeCN)2 in dichloromethane at –100°C. [B12H12]2– → [B12H11SeCN]2–
- 10. Applications: ⸙ Closo-dodecaborate anion takes advantage of high stability under usual conditions that allows to use its salts as components of high burning composite propellants. ⸙ An important field is using lithium closo-dodecaborates Li2[B12H12] and Li2[B12Cl12] as used for advanced rechargable lithium batteries. ⸙ The main direction of application of the closododecaborate derivatives nowadays is medicine. ⸙ [B12H12]2-, [B12(OH)12]2- and [B12(OMe)12]2- show promise for use in drug delivery. ⸙ Salts of dodecaborane is therapeutic agent in cancer treatment.
