Chapter6 140330084152-phpapp02
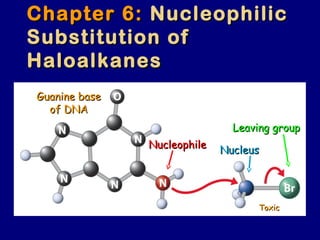
- 1. Chapter 6:Chapter 6: NucleophilicNucleophilic Substitution ofSubstitution of HaloalkanesHaloalkanes Guanine baseGuanine base of DNAof DNA Leaving groupLeaving group NucleusNucleusNucleophileNucleophile ToxicToxic
- 2. HaloalkanesHaloalkanes NamesNames:: Halo-Halo-, as a substituent, as a substituent 1-Chlorobutane1-Chlorobutane (1(1SS,2,2RR)-1-Bromo-2-)-1-Bromo-2- fluorocyclohexanefluorocyclohexane 2-Iodo-2-2-Iodo-2- methylbutanemethylbutane CC ++ -- XX ClCl 44 33 22 11 FF BrBrRR SS II CHCH33
- 3. The C-X Bond isThe C-X Bond is PolarizedPolarized ClClCCHH33 ++ -- ElectrophilicElectrophilic
- 5. Trends are a function of dipole-Trends are a function of dipole- dipole and London forcesdipole and London forces ElectronegativityElectronegativity (4.0)(4.0) (3.2)(3.2) (3.0)(3.0) (2.7)(2.7)
- 6. Nucleophilic Substitution:Nucleophilic Substitution: GeneralGeneral Color scheme:Color scheme: NuNu,, EE,, LL,, andand curved arrowscurved arrows: e-Flow: e-Flow CC XXNuNu ++ CC XXNuNu -- ++ NucleophileNucleophile (Nu)(Nu) LeavingLeaving group (L)group (L) ElectrophileElectrophile (E)(E) --
- 7. Remember: Acid-base reactionsRemember: Acid-base reactions BB ++ HH AA BB HH ++ AA -- -- conjugateconjugate acid baseacid base -- BB == NuNu -- wwhenhen HH is attacked, we call itis attacked, we call it base B.base B. WhenWhen CC (or other nuclei) attacked, we call it(or other nuclei) attacked, we call it nucleophile Nu.nucleophile Nu.
- 8. Note: no (simple) ∆H º calculations possible on ionic reactions; bond strengths refer to homolytic, not heterolytic, dissociation.
- 9. Note: Tertiary halides are notably absent from this list.Note: Tertiary halides are notably absent from this list.
- 10. MechanismMechanism In general: how do we study it ?In general: how do we study it ? 1.1. KineticsKinetics 2.2. StereochemistryStereochemistry 3.3. Modify substituents: look for electronic andModify substituents: look for electronic and steric effectssteric effects 4.4. Isotope effects: Usually H/DIsotope effects: Usually H/D DHºDHº CC----HH << CC—D—D 5.5. Modify reagents/subtrates: Nu, E, L, solventModify reagents/subtrates: Nu, E, L, solvent
- 11. KineticsKinetics ForFor HOHO ++ CCHH33 ClCl CHCH33OH +OH + ClCl ---- Rate =Rate = kk [CH[CH33Cl][ OH]Cl][ OH] 22ndnd orderorder Points toPoints to bimolecularbimolecular mechanism (TS)mechanism (TS) Hence name:Hence name: SSNN22 bimolecular,bimolecular, nucleophilic substitutionnucleophilic substitution --
- 12. [HO[HO······CHCH33Cl]Cl]‡‡ ?? CHCH33ClCl CHCH33OHOH EE What is TSWhat is TS structure?structure? We can look atWe can look at stereochemistrystereochemistry.. Two extreme approaches ofTwo extreme approaches of NuNu :: CC XX BackBack FrontFront + Cl+ Cl-- ++ -- OHOH Transition StateTransition State ――
- 13. FrontsideFrontside attack:attack: RetentionRetention ofof configuration.configuration. BacksideBackside attack:attack: InversionInversion ofof configuration.configuration. Which one is it?Which one is it? CC XX chiralchiral ** Test: UseTest: Use enantiomerically pureenantiomerically pure
- 16. CC BrBr SS HH HH33CC CHCH33CHCH22 ++ -- II CC BrBrII -- ++ HH CHCH33 CHCH22CHCH33 RR Result:Result: InversionInversion ((nono SS -product)-product)
- 18. Chemical Consequences ofChemical Consequences of InversionInversion 1. Retention: By double inversion1. Retention: By double inversion CC CHCH33 RR HH BrBr CC CHCH33 RR HH SHSHCCII CHCH33 RR HH II -- BrBr -- II -- H SH S -- ++ -- -- ++ 2. Inversion does not necessarily mean:2. Inversion does not necessarily mean: R SR S CHCH33CHCH22OO ++ CC CHCH33SS HH HH33CC BrBr CC XX CHCH33CHCH22OO --++ SCHSCH33 HH CHCH33 SS cc bb aabb cc aa-- SS
- 19. 3. Diastereoisomerization3. Diastereoisomerization CHCH33 BrBr HH CHCH33 CHCH33CHCH22 HH CHCH33 HH HH CHCH33 CHCH33CHCH22 IIII -- BrBr RR RR RR SS -- -- BrBr HH HH33CC HH SSSS HH CNCN HH33CC HH RRSSCNCN -- BrBr -- -- CHCH33 BrBr RR SS II -- CHCH33 RR RR II CisCis TransTrans -- BrBr--
- 20. Leaving Group AbilityLeaving Group Ability “L”“L” (kinetic parameter)(kinetic parameter) CC LLNuNu ++ BB ++ HH AA -- What makes a goodWhat makes a good LL-- ((AA-- ) ?) ? Remember from the discussion on acidity:Remember from the discussion on acidity: 1.1. Ability toAbility to accommodate e-pairaccommodate e-pair (charge) :(charge) : e-Negativity + resonancee-Negativity + resonance 2.2. Size of the orbitalSize of the orbital describing the e-pair.describing the e-pair. 3.3. Indirectly:Indirectly: Bond strengthBond strength CC——LL ((HH——AA))
- 21. F < Cl < Br < IF < Cl < Br < I Increasing,Increasing, goinggoing downdown periodicperiodic table (PT).table (PT). HF HCl HBr HIHF HCl HBr HI ppKKaa 3.2 -2.2 -4.7 -5.23.2 -2.2 -4.7 -5.2 DDHºHº 135 103 87 71135 103 87 71 Why?Why? Because:Because: GoesGoes downdown in PTin PT And:And: Orbital sizeOrbital size increasesincreases from 2p to 3p to 4p…from 2p to 3p to 4p… As noted earlier: This trend isAs noted earlier: This trend is oppositeopposite thatthat expected on the basis ofexpected on the basis of electronegativityelectronegativity (goes(goes down in PT).down in PT). SameSame trend astrend as HX:HX: -- -- -- -- Halides asHalides as LL
- 22. ppKKaa 50 35 15.7 3.250 35 15.7 3.2 DDHºHº 105 107 119 135105 107 119 135 increasesincreases In practice: onlyIn practice: only FF is a reasonableis a reasonable leaving groupleaving group inin this row. Hydroxide can be, in special cases.this row. Hydroxide can be, in special cases. -- CHCH33<< NHNH22<< OHOH<< FF :: ElectronegativityElectronegativity wins !wins ! decreasesdecreases,, butbut And:And: SizeSize of orbitalof orbital decreasesdecreases.. RR LL oror HH AA:: Along a row of PT:Along a row of PT: LL increasesincreases to theto the rightright (same trend as acidity)(same trend as acidity)
- 23. For example, for same leaving atom, e.g.,For example, for same leaving atom, e.g., Generally:Generally: LL increasesincreases to theto the rightright andand downdown PT.PT. But, superimposed on these trends:But, superimposed on these trends: Resonance.Resonance. ppKKaa :: 15.5 4.7 -1.215.5 4.7 -1.2 -- -- -- OO OO OO -- CHCH33OO << CHCH33COCO << CHCH33S OS O RORO :: (of acid)(of acid)
- 24. Neutral LNeutral L areare goodgood: relatively: relatively nonbasicnonbasic 1. Protonated alcohols:1. Protonated alcohols: L = HL = H22OO Use ROH plus HBr, or HI, or HUse ROH plus HBr, or HI, or H22SOSO44 2. Diazonium ions:2. Diazonium ions: L = NL = N22 , a superleaving group, a superleaving group RR OOHH ++ HH RR OO HH HH NuNu--++ RR NuNu ++ HH22OO RR N NN N ++ NuNu RR NuNu ++ N NN N --++ ++
- 25. For practical purposes:For practical purposes: Here is whereHere is where LL-- abilityability ends (going down PT)ends (going down PT)
- 26. Nucleophilicity “Nu” (kinetic parameter) Affected by charge, basicity, solvent,Affected by charge, basicity, solvent, polarizability, sterics.polarizability, sterics. 1.1. ChargeCharge (for same atom):(for same atom): TheThe moremore charged, thecharged, the moremore nucleophilicnucleophilic HO > HHO > H22O ; HO ; H22N > HN > H33N ; SON ; SO44 > ROSO> ROSO33 -- -- 2-2- --
- 27. 2.2. BasicityBasicity:: DecreasesDecreases to the rightto the right in PT, so doesin PT, so does NuNu:: HH33N > HN > H22O ; HO ; H22N > HO ; HO > FN > HO ; HO > F -- -- -- -- Comparison of neutral and chargedComparison of neutral and charged NuNu: (See: (See ppKKaa table)table) HH22N > HO > HN > HO > H33N > F > HN > F > H22OO -- -- -- As expected: TrendAs expected: Trend oppositeopposite LL abilityability
- 28. ProticProtic solventssolvents havehave acidic Hacidic H ; e.g., RO; e.g., RO HH oror NN HH. They. They surroundsurround chargedcharged NuNu , using, using hydrogen bonds:hydrogen bonds: ++ ++-- ++-- -- -- RR Down the PT:Down the PT: Murky!Murky! BasicityBasicity goesgoes downdown,, hence Nu should too (opposite L). But nothence Nu should too (opposite L). But not true: Nutrue: Nu increasesincreases!! The reason:The reason: SolventSolvent effects andeffects and polarizabilitypolarizability (deformability of orbital of(deformability of orbital of ) have a strong influence.) have a strong influence. For chargedFor charged NuNu-- :: NuNu NuNu HH OROR
- 29. Protic Solvents:Protic Solvents: Fluoride is aFluoride is a WorseWorse Nucleophile Than IodideNucleophile Than Iodide HydrogenHydrogen bondsbonds δ- δ- δ- δ- δ- δ+ δ+ δ+ δ+ Solvent shell increases “size” ofSolvent shell increases “size” of NuNu ,, hencehence NuNu increasesincreases goinggoing downdown PT.PT. --
- 30. For unchargedFor uncharged NuNu ,, samesame trend, buttrend, but now due tonow due to polarizabilitypolarizability HH22O < HO < H22S < HS < H22Se (CHSe (CH33))33N < (CHN < (CH33))33PP Polarizability operates also forPolarizability operates also for charged Nu, which already benefitcharged Nu, which already benefit from lesser solvation: especially fast.from lesser solvation: especially fast.
- 31. IncreasingIncreasing PolarizabilityPolarizability ImprovesImproves NucleophilicityNucleophilicity More polarizableMore polarizable Less polarizableLess polarizable
- 32. The Story Changes In Polar Aprotic Solvents, Which • dissolve salts • do not form H bonds • enable formation of “naked” anions cause huge rate increases with all Nu follow trends in basicity
- 34. Review: Range ofReview: Range of NucleophilicitiesNucleophilicities • charge • basicity • polarizability • H-bonding Depends on:Depends on:
- 35. Summary Trends in theSummary Trends in the Periodic TablePeriodic Table L Nu- “Naked” anions, aprotic solvents or Protic solvent, polarizability e-Negativity > DHº and orbital size e-Negativity < DHº and orbital size
- 36. Steric EffectsSteric Effects Sterics forSterics for LL:: LargerLarger == betterbetter Sterics forSterics for NuNu:: LargerLarger == worseworse,, e.g., CH3O > (CH3)3CO -- -- Sterics aroundSterics around EE are theare the mostmost significant.significant.
- 37. StericsSterics aroundaround electrophilicelectrophilic CC LL RR Br + IBr + I RR I + Br relative ratesI + Br relative rates CHCH33 CHCH33CHCH22 (CH(CH33))22CHCH (CH(CH33))33CC 145 1 0.078 0145 1 0.078 0 MechanismMechanism changeschanges CHCH33CHCH22 CHCH33CHCH22CHCH22 (CH(CH33))22CHCHCHCH22 (CH(CH33))33CCHCCH22 1 0.8 0.031 0.8 0.03 slow!slow! 1010-5-5 -- -- αα :: ββ :: AlphaAlpha versusversus betabeta branching:branching:
Editor's Notes
- 1:45SwingBrotherSwing