Research statement dr. khan
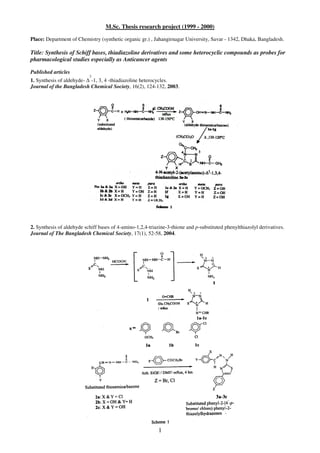
- 1. 1 M.Sc. Thesis research project (1999 - 2000) Place: Department of Chemistry (synthetic organic gr.) , Jahangirnagar University, Savar - 1342, Dhaka, Bangladesh. Title: Synthesis of Schiff bases, thiadiazoline derivatives and some heterocyclic compounds as probes for pharmacological studies especially as Anticancer agents Published articles 1. Synthesis of aldehyde- ∆ 2 -1, 3, 4 -thiadiazoline heterocycles. Journal of the Bangladesh Chemical Society, 16(2), 124-132, 2003. 2. Synthesis of aldehyde schiff bases of 4-amino-1,2,4-triazine-3-thione and p-substituted phenylthiazolyl derivatives. Journal of The Bangladesh Chemical Society, 17(1), 52-58, 2004.
- 2. 2 UNESCO - IUPAC research project (Jul’2004 - Oct’2005) Place: Institute of Macromolecular Chemistry, Academy of Science of the Czech Republic, Prague 6. Topic: Functionalized bisphosphonic acids for surface modification of magnetic iron oxide and other metal oxides EUROGREENPOL- In book of proceedings p.27-32. First european summer school on green chemistry of polymers, Iasi, Romania, 21-27 August 2005. Abstract The aim of this work was to modify the surface of magnetic oxide nanoparticles such as magnetite- Fe3O4, maghemit-γFe2O3, ferrites MeO.Fe2O3 where Me: Cu, Co, Mn. Recently modified magnetic oxides have been reported to have wide range of applications like immobilization of biopolymers (antibodies, enzymes, proteins, nucleic acids etc), magnetic resonance imaging, bone and tissue repair (as bisphosphonates have high affinity for hydroxyapatite), cancer treatment by hyperthermia, site-specific delivery of drugs, gene delivery to cells and so on. To fullfill these properties we synthesized and characterized various kinds of bisphosphonates (I - III) with additional functional groups such as -NH2, OH, CH=CH2, -NH-NH2, halogen, PEG, polymer etc. in the molecules. A. Direct synthesis of 1,1-bisphosphonate (type I) Examples: H2N(CH2)6COOH, Br(CH2)6COOH etc. The aim of the above synthesis is to introduce H2N-NH (hydrazine) group at the end of bisphosphonic acid which is important for immobilization of biopolymers, chelating group for specific sorption of cations etc. CH2=CHCOOC2H5 H2NNH2.H2O+ reflux,5.5 hrs N NO H H EtOH aq.KOH soln.i. ii. neutralize, Conc.HCl H2NNHCH2CH2COOH 3-hydrazinopropionic acid Pyrazolidin-3-one OH OH P O OH OH P O OH OH OH P O OH OH P O R3H OH OH P O N OH OH P O R1 R2 I II III R(CH2)nCOOH PCl3, H3PO3 reflux, 70C, 21 hrs CH3SO3H i. ii. R(CH2)n C P(O)(OH)2 P(O)(OH)2 OH H2O (type I)
- 3. 3 B. Michael addition reaction of ethylenebisphosphonate (type II) C. Direct synthesis of α-aminophosphonic acid (type III): Mannich-type reaction D. Incorporation of bisphosphonate moiety in the polymer chain: free radical reaction H2C CH[P(O)(OH)2]2 N(H2CCH2OH)2 H2C=C[P(O)(ONa)2]2 HN(CH2CH2OH)2+ CH3COOH, H2O reflux,110-120C, 11hrs i. ii. Cation exchanger, 2(N)H2SO4 H2C=C[P(O)(ONa)2]2 H2C=C[P(O)(OH)2]2 H2C=C[P(O)(OCH3)2]2 H2C CH[P(O)(OCH3)2]2 N(H2CCH2OH)2 H2C CH[P(O)(OK)2]2 N(H2CCH2OH)2 H2C CH[P(O)(OH)2]2 N(H2CCH2OH)2 Cation exchanger excess HC(OCH3)3 relux, 60-65 C and then 106 C HN(CH2CH2OH)2 relux, 65-76 C,11 hrs KOH, EtOH relux, 85C, 6hrs Cation exchanger Example: CH2=CHCH2NH2 ; R N CH2 CH2 P(O)(OH)2 P(O)(OH)2 RNH2 + aq.HCHO +H3PO3 Conc.HCl, H2O reflux,105C,3.5 hrs O2N.C6H5NH23 H2N O SO3H HN[CH2P(O)(OH)2]2+ reflux, 100-107C, 2 hrs H2O H2NN[CH2P(O)(OH)2]2 OH OH HO HCHO ; HN[CH2P(O)(OH)2]2 reflux,73-78C, 2hrs CH3COOH ; H2O OH OH HO CH2N[CH2P(O)(OH)2]2 Pyrogallol (type III) (type II) (type II) H2C CH N[CH2P(O)(OH)2]2CH2 CH2=CHCONHCH(CH3)2CH2=C[P(O)(ONa)2]2 CH2=C[P(O)(OH)2]2 i. ii. iii. (NH4)2S2O8, 75C, 24 hrs Block copolymer of NIPAAm and the corresponding diphosphonic acid i. CH2=C[P(O)(ONa)2]2 H2C CH N[CH2P(O)(OH)2]2CH2 PEG 4000 Ce(iv)(NH4)NO3 N2 atm,30 C, 3 hrs Block/graft copolymer of PEG and the corresponding diphosphonic acid ii.
- 4. 4 Terminology: PEG 4000 = Polyethylene glycol( mol. Wt . 4000) NIPAAm = N-Isopropylacrylamide HN[CH2P(O)(OH)2]2 = imidodi(methylenephosphonic acid) H2N-O-SO3H = hydroxylamine-O-sulfonic acid HN(CH2CH2OH)2 = diethanolamine Ce(iv)NH4NO3 = Ceric ammonium nitrate (used as redox initiator) (NH4)2S2O8 = Ammonium persulfate (used as free radical initiator) HC(OCH3)3 = trimethyl orthoformate H2NNH2.H2O = Hydrazine monohydrate H2N(CH2)6COOH , Br(CH2)6COOH = 6-amino / bromo caproic acid References [1] G.R Kieczykowski. J.Org.Chem. 1995, 60 (25), 8310-8312. [2] M. Lecouvey, Heteroatom Chem. 2000, 11(7), 556-561. [3] Sergio H. Szajnman. Bioorg. Med. Chem. Lett. 2001, 11, 789-792. [4] D. Allan Nicholson, W.A. Cilley and O.T. Quimby. A convenient method of esterification of Polyphosphonic acids. J.Org. Chem.1970, 35(9), 3149-3150. [5] Charles R. Degenhardt and Don C.Burdsall. Synthesis of Ethylidenebis(phosphonic acid) and its tetraalkyl esters. J.Org.Chem. 1986, 51(18), 3488-3490. [6] David W. Hutchinson and David M. Thornton. Michael addition reactions of ethenylidenebisphosphonates. J. Organomet. Chem. 1988, 346, 341-348. [7] Kurt moedritzer and Riyad R. Irani. The direct synthesis of α-aminomethylphosphonic acids. Mannich type reactions with orthophosphoric acid. J.Org.Chem. 1966, 31, 1603-1607. [8] Amination of Tertiary Amines by Hydroxylamine-O-sulfonic Acid. J. Org. Chem. 1959, 24, 859. [9] Synthesis of Block copolymers via redox polymerization. J. Appl. Polym. Sci. 1999, 71, 1385-1395. [10] Shigeo Nakamura, Masato Amano, J. Polym. Sci., Part A: Polym. Chem., 1997, 35, 3359-3363. 4. Synthesis of 1,1′-binaphthalene-2,2′-diamine based ligand for enantioselective recognition of chiral carboxylate anions NH2 NH2 CF3 CF3OCN N H N H O O N H N H CF3 CF3 CF3 CF3 1, 2, 3 + 1. Dry CH2Cl2, 28°C, 20 h, 2. quench with distilled methanol, stir 20 h 3. column chromatography P.E: DCM, 1:1, 100% DCM (R)-(+)-1,1-binaphthalene -2,2- diamine 3,5-bis(trifluoromethyl) -phenylisocyanate (R)-2,2′-Bis{N-[3,5-bis(trifluoromethyl) phenyl]ureido}-1,1′-binaphthalene
- 5. 5 ABSTRACT - Ph.D Thesis (Sept, 2006 – Jul, 2011) Place: Department of Chemistry, Masaryk University in Brno, Czech Republic (EU). Ph.D thesis title: Modified cucurbit[n]urils: synthesis and supramolecular interactions There has been a remarkable development in the cucurbit[n]uril (CBn, n = 5-10) chemistry since the last decade. The development outcomes also inspired us to contribute efforts to this rapidly growing topic. The very common feature of the parent CBn and their homologues is the limited solubility or insolubility in common solvents (except in aqueous acid media) which limits their applications. That’s why modification of the parent CBn with suitable substituents is required to facilitate their solubility in solvents like water, DMSO, methanol, acetonitrile etc. Figure 1. Unsubstituted CB6 (n = 6) First project of this thesis was dealt with the syntheses and characterizations (1 H NMR, 13 C NMR and MALDI-TOF MS) of a series of monosubstituted CB6 having a substitutent at the methylene bridge (axial) position of CBn by reacting glycoluril with various aldehydes (RCHO). I developed new purification method for a monosubstituted CB6 substituted at the methylene bridge position (Figire 2. I-01). Figure 2. Monosubstituted CB6 (n = u+s = 5+1 = 6) Second project was dedicated to the synthesis, improved purification, characterization and to study supramolecular properties of a robust water soluble macrocyclic receptor, hexamethylcucurbit[6]uril (MeCB6). Good solubility of MeCB6 in pure water allowed us to study its supramolecular interactions with the synthesized guests II.1 - II.10 using sophisticated techniques like FT-NMR, UV-vis spectrophotometry, VP-ITC, MALDI-TOF MS and X-ray N N NN O O N N NN O O C H C H2 5 1 I-01
- 6. 6 diffraction. We determined the binding modes of these guests in solution using 1 H-NMR technique and studied elaborately the effect of NaCl on the binding affinities (K /M-1 ) of MeCB6 with them. Binding modes of MeCB6 with guests in the solid state were studied by preparing new X-ray quality single crystals of several inclusion complexes of MeCB6 with the guests. NH N NH3 NH3 NH3 NH3 NH3 NN NN NN N N H3 N NH3 I2 I2 I2 I2 -. N N CO2 HHO2 C NH3 H3 N .2Cl - + + NH3 -.Cl + . - I N N N N I2 + II.3a II.3b II.3c II.4 II.5 II.6a II.6d II.6b II.6c II.6e II.7 II.8 II.9 1 6 71 1 8 1 2 4 1 2 4 1 2 4 2 2 1 1 4 4 + + + + + -.Cl + + ++ + + + + + + -. -. -. -.Br -.Cl -.Cl -.Cl -.Cl - .2Cl II.10 ++ - .2Br II.2 + II.1 ++ -. Figure 3. Structure of hosts (CB6, CB7 and MeCB6) and guests (II.1 - II.10) used in the study. Published articles 1. V. Kolman, M. Shamsul Azim Khan, M. Babinsky, R. Marek, V. Sindelar. Supramolecular Shuttle Based on Inclusion Complex between Cucurbit[6]uril and Bispyridinium Ethylene. Organic Letters, 13 (23), 6148-6151, 2011. (IF 5.862, 2011) 2. M. Stancl, M. Shamsul Azim Khan, V. Sindelar. 1,6- Dibenzylglycoluril for Synthesis of Deprotected Glycoluril Dimer. Tetrahedron, 67, 8937-8941, 2011. (IF 3.025, 2011) 3. M. Shamsul Azim Khan, D. Heger, M. Necas, V. Sindelar. Remarkable salt effect on stability of supramolecular complex between modified cucurbit[6]uril and methylviologen in aqueous media. Journal of Physical Chemistry B, 113 (32), 11054-11057, 2009. (IF 4.19, 2009) CB6 CB7 MeCB6
- 7. 7 Post-doctoral research project (Aug, 2011 to Dec, 2012) Place: Institute of Organic Chemistry and Biochemistry, Academy of Science of the Czech Republic, Prague 6. Project Title: Synthesis of cyclodextrin heteroduplexes and study of their supramolecular properties Note: Results will be published after gathering some more results.
- 8. 8 Synthesis of the compounds 5 and 6 from the starting reactant α-cyclodextrin (α-CD): 6 steps O OH HO OH O O OH OH OH O O OH OH HO O O O OH OH HO O OH HO HO O O OHHO OH O O OBn BnO OBn O O OBn BnO OBn O O OBn OBn BnO O O O OBn OBn BnO O OBn OBn BnO O O OBnBnO OBn O O OBn BnO OH O O OBn OBn OBn O O OBn OBn BnO O O O OBn OBn HO O OBn BnO BnO O O OBnBnO OBn O O OBn BnO Br O O OBn BnO OBn O O OBn OBn BnO O O O OBn OBn Br O OBn OBn BnO O O OBnBnO OBn O O OH HO Br O O OH OH OH O O OH OH HO O O O OH OH Br O OH HO HO O O OHHO OH O O OH HO SCOCH3 O O OH OH OH O O OH OH HO O O O OH OH H3COCS O OH HO HO O O OHHO OH O O OH HO SH O O OH OH OH O O OH OH HO O O O OH OH HS O OH HO HO O O OHHO OH O 1 23 4 5 6 α-CD Compound 5 ≡ a c d e f b
- 9. 9 Reaction conditions: [a] i. NaH, BnCl, dry DMSO, Ar atm, RT, 18h, ii. hydrolysis with MeOH; [b] i. DIBAL-H, 50°C, Ar atm, ii. hydrolysis; [c] CBr4, Ph3P, DMF, 60°C; [d] Pd/C, 40 bar, DMF- EtOH, RT; [e] CH3COSK, DMF, RT; [f] 1 M NaOH, MeOH-H2O, RT. Synthesis of the compound 11 from the starting reactant β-cyclodextrin (β-CD): 5 steps Purification method Compounds 1-6 (for α-CD series) and 7-11 (for β-CD series) were purified using flash chromatography (compounds 1-3 and 7-9) in acetone-toluene mobile phase and reverse phase chromatography (compounds 4-6 and 10-11) in methanol-water mobile phase. β-CD 11 Compound 11 ≡ a - e
- 10. 10 Synthesis of αβCD- heteroduplex from the compounds 5 and 11: one pot synthesis Reaction conditions: [g] i. HPLC water, ii. 1 M NaOH, stir at RT for 1 d ; iii. NaHCO3-Na2CO3 aq. buffer (pH = 9), air oxidation, 5 d. O OH HO OH O O OH HO SCOCH3 O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH H3COCS O O OH HO HO O O OH HO SCOCH3 O O OH OH OH O O OH OH HO O O O OH OH H3COCS O OH HO HO O O OHHO OH O 11 5 α-CD β-CD αβCD - heteroduplex O OH HO S O O OH OH OH O O OH OH HO O O O OH OH S O OH HO HO O O OHHO OH O O OH HO OH O O OH HO S O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH S O O OH HO HO O α-CD β-CD O OH HO S O O OH OH OH O O OH OH HO O O O OH OH S O OH HO HO O O OHHO OH O O OH HO S O O OH OH OH O O OH OH HO O O O OH OH S O OH HO HO O O OHHO OH O ααCD - homoduplex α-CD α-CD 12 13 O OH HO OH O O OH HO S O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH S O O OH HO HO O O OH HO OH O O OH HO S O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH S O O OH HO HO O ββCD - homoduplex Two isomers (ββ1 and ββ2) β-CD β-CD 14a-b O OH HO SO O OH OH OH O O OH OH HO O O O OH OH S O OH HO HO O O OHHO OH O O OH HO OH O O OH HO SO O OH OH OH O O OH OH OH OO OH OH HO O O OH OH S O O OH HO HO O βCD-intramolecular dithiolαCD-intramolecular dithiol α-CD β-CD 16 15 g
- 11. 11 Compounds 12-16 were separated and purified by Reverse Phase Chromatography with gradient elution using MeOH-H2O mobile phase system. The isolated compounds were then analyzed by HPLC using both ELSD and UV detector. The compounds were characterized by 1 H NMR, MALDI-TOF MS and HR-MS techniques, elemental analysis etc. Template assisted synthesis of αβCD- heteroduplex from the compounds 5 and 11 O OH HO OH O O O H HO SCOCH3 O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH H3COCS O O OH HO HO O O OH HO SCOCH3 O O OH OH OH O O OH O H HO O O O O H OH H3COCS O OH HO HO O O OHHO OH O 115 O OH HO S O O OH OH OH O O OH OH HO O O O OH OH S O OH HO HO O O OHHO OH O O OH HO OH O O OH HO S O O OH OH OH O O OH OH OH OO OH OH HO O O OH OH S O O OH HO HO O α-CD β-CD Template that showed strong binding affinity (K = 107 M-1 ) with the αβCD- heteroduplex (binding affinity was prior modelled using computation and then experimentally measured by isothermal titration calorimetry) 13
- 12. 12 Determination of Association Constants (K / M-1 ) between various guests and duplex 13 in aqueous medium using VP-ITC (isothermal titration calorimetry) technique As guest molecules few organic dyes, fluorescent probe and anticancer drug were used to determine their binding affinity with the duplex 13. Computation modelling (using Vina autodock programme) was used to study the possible binding mode and affinity of each guest inside the 13 cavity before experimental determination of the binding affinity. References: 1. The Journal of Organic Chemistry 74: 1082-1092, 2009. 2. Chemistry - A European Journal 18: 12292-12304, 2012.
