Ophthalmic preparations
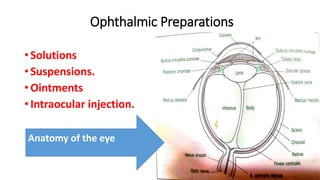
- 1. Ophthalmic Preparations • Solutions • Suspensions. • Ointments • Intraocular injection. Anatomy of the eye
- 2. Ophthalmic preparations should be sterile unlike otic or nasal Not necessary to be isotonic, Explain?
- 3. Advantages • Ensures that the required site. • Low incidence of side effects. • Administered easy by the patient.
- 4. Disadvantages • Low retention time; absorption of 1% of the original dose. • Loss of the majority of dose by tears; two drops is 30 µL and the tears volume is 7 µL- 30 µL. • Patient inconvenience; pain, irritation and blurring of vision. • Being sterile requires special facility for manufacturing.
- 5. Formulation of Aqueous Ocular Dosage Form Drug salt. Physical properties of the dispersed phase of the therapeutic substance. pH Chemical stability Absorption across the cornea Vehicle Viscosity Preservative Antioxidant Surface active agent.
- 6. Choice of the drug salt • Solubility - Pain and irritation ( stinging) - Adrenaline bitartarate pH 3-4 moderate stinging - Adrenaline borate pH 5.5 – 7.5 mild stinging - Adrenaline hydrochloride pH 2.5 – 4.5 neutralized Reduced stingin • Salt producing discomfort will result in lacrimation anfd hence the wash effect.
- 7. Physical Properties of dispersed agent • Particle Size: - large particles causes irritation • 95% of the dispersed particles should have average particle diameter less than 10 µm.
- 8. pH • Ideally should be 7.4 and this the tears pH. • This to enhance stability, acceptability and absorption.
- 9. Chemical Stability • Effect of pH and temperature on the stability of pilocarpine Temp pH Half-life 121 6.8 34 min 121 5 24 hrs 25 6.8 66 days 25 5 Several years
- 10. Drug Absorption Through the Cornea • Co-existance of the drug in ionized and non-ionized to achieve effective absorption due to the different nature of the corneal layers. Layer Nature Drug form Epithellium Lipid rich Non-ionized Stroma Aqueous Ionized endothellium Lipid rich Non-ionized
- 11. Vehicle •Purified water Not WFI is commonly used. USP
- 12. Viscosity • Viscosity modifier is required to: 1- control the drop unit from the container. 2- to control the residence time of the solution within the precorneal. • 55 mP/s above which no increase in the contact time. • Formulation should be less than 30 mP/s • Increasing the viscosity will enhance the physical stability of the suspension.
- 13. • The viscosity modifier should have the following properties: - Easily filtered through 0.8 micron filter - Easily sterilized by filtration or heat. - Compatible with the components. Examples of viscosity modifiers: - HPMC 0.45 – 1% w/w - Poly vinyl alcohol 0.25 – 3% w/w - Polyacrylic acid.
- 14. Preservative because it is multi-dose container • Benzalkonium chloride 0.01% w/v. to enhance its activity agaist pseudomonas aurignosa we added EDTA 0.1%. • Organic mercurial compounds 0.001-0.002% w/v but of limited use. • Organic alcohols: chlorobutanol ( 0.5% w/v)and phenylethylalcohol 0.25 – 0.5% w/v. Cationic preservative
- 15. Antioxidants: to optimize the stability of therapeutic agent that degrade by oxidation as sod.metabisulphite 0.3% • Surfactants: Non-ionic sufactants are generally used.
- 16. Manufacture of Aqueous ophthalmic Formulations + Similar to parenteral preparations. • Cleanroom conditions followed by sterilization by autoclaving. • Clean or aseptic conditions followed by aseptic sterilization by filtration 0.22 micron. • Production under aseptic condition; dispersion of sterile therapeutic agent into sterile vehicle and excipients and packing under aseptic conditions suitable for suspension.
- 17. Ophthalmic Ointments • Dispersion of therapeutic agent into prepared ointment base in the same way as general ointments but required to be sterile. • Have a disadvantage that it causes greasiness and blurring of vision but it can be used during night time.
- 18. Ointment Bases for Ocular Administration 1- Hydrocarbon bases: mixture of paraffins ( yellow soft paraffin, white soft paraffin) 2- Non-emulsified absorption bases: liquid and yellow soft paraffins and emulsifying agent. lanolin derivatives; aqueous solution can be incorporated. 3- water soluble bases/ aqueous gels; polyethylene glycol, polyacrylic acid gels
- 19. Other excipients • Non-aqueous antioxidants butylated hydroxyl toluene, butylated hydroxyanisole.
- 20. Manufacture of Ophthalmic Ointment • Same as ointments but required to be sterile . • Manufactured and packed under aseptic conditions. • Sterile therapeutic agent added to sterile base> • Filling in container under aseptic conditions.
- 21. Nasal Formulations • Aqueous based system • Rapid absorption • Avoid first pass metabolism
- 22. • Control of pH: 5.5 – 6.5 • Tonicity : Inclusion of NaCl • Choice of vehicle: Purified water, non-aqueous vehicles are not used. Glycerol can be added as co-solvent and humectant. • Viscosity modifier: to enhance administration and retention of drops on the nasal mucosa. methylcellulose, hydroxyethylcellulose, polyacrylic acid. • Preservative: chlorobutanol 0.5%, Parabens 0.2% benzalkonium chloride 0.003 -0.02% w/v. • Antioxidants: Sod.metabisulphite, Sod.sulphite. • Manufacture: similar to non-sterile liquid preparations.
- 23. Otic Formulations • May be aqueous or non-aqueous. • Vehicles: depends on the solubility of the active substance. - Aqueous vehicle as purified water. - Non-aqueous as mineral oils as paraffin oil, vegetable oils as archis oil. - Non-aqueous but miscible with water as glycerol. Preservative, antioxidants and viscosity modifier can be added.