Cold chain system for vaccines
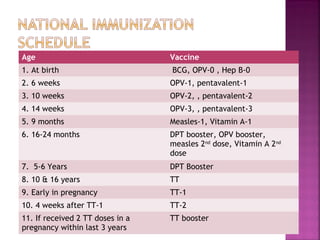
- 1. Age Vaccine 1. At birth BCG, OPV-0 , Hep B-0 2. 6 weeks OPV-1, pentavalent-1 3. 10 weeks OPV-2, , pentavalent-2 4. 14 weeks OPV-3, , pentavalent-3 5. 9 months Measles-1, Vitamin A-1 6. 16-24 months DPT booster, OPV booster, measles 2nd dose, Vitamin A 2nd dose 7. 5-6 Years DPT Booster 8. 10 & 16 years TT 9. Early in pregnancy TT-1 10. 4 weeks after TT-1 TT-2 11. If received 2 TT doses in a pregnancy within last 3 years TT booster
- 2. Cold chain system for vaccines
- 3. The Cold Chain • The "cold chain" is a system of storage and transport of vaccines at low temperature from the manufacturer to the actual vaccination site. • The cold chain system is necessary because vaccine failure may occur due to failure to store and transport under strict temperature controls.
- 4. Why have the Cold Chain? If vaccines are exposed to excessive They lose their potency Heat Light Freezing
- 5. In general • All Vaccines lose potency on exposure to heat above +80 C • Some Vaccines lose potency when exposed to freezing temperatures • The damage is irreversible
- 6. Cold chain Equipment • Walk in freezers • Walk in coolers (WIC) • Deep freezers • ILR-Basket • Dial Thermometer • Cold boxes • Vaccine carriers • Day carriers
- 7. Cold Chain Equipment: Various Levels –State Refrigerator Truck Walk-in Freezer Walk-in Cooler
- 8. Cold Chain Equipment: District Level Cold Box Deep Freezer (DF) 300 ltr Ice-Lined Refrigerator(ILR) 300 ltr
- 9. Cold Chain Equipment: Unit Level Ice-Lined Refrigerator(ILR) 140 ltr Deep Freezer (DF) 140 ltrCold BoxVaccine Carriers
- 10. 1. Walk-in Cooler & Freezer Walk-in Cooler (+2o C to +8o C) Walk-in Freezer (-15o to -25o C)
- 11. Walk-in-Freezers (WIF) • used for bulk storage of OPV, and also to prepare frozen ice packs at state stores. • Maintain a temperature around (-) 20oC. • available in sizes of 16.5 Cum. and 32 Cum. • Provided with two identical cooling units and standby generator. • installed in all of the states • Serves 4-7 districts
- 12. Walk-in-Coolers (WIC) • used for bulk storage of vaccines at State and Regional/Divisional Stores. • maintain a temperature of +2oC to +8oC. • Available in sizes of 16.5 Cum. and 32 Cum. • used for storage of large quantities of vaccines, like DPT, DT, TT, Measles, BCG, Hepatitis B • also provided with temperature recorder and alarm • WIC/WIF store vaccines of three months requirement and 25% buffer stock for the districts they cater. • Cater 4-5 districts • 3 deep freezers and 10 cold boxes.
- 13. • Vaccine delivery vans • Transportation of vaccines from Regional centers to districts • From districts to PHCs • Temperature maintained at +2o to +8o C. 2. Refrigerated Truck
- 14. 3. Ice-Lined Refrigerator (ILR) Size: 300 ltr / 140 ltr Level: District (300 ltr) / PHC (140 ltr) Temperature: +2˚C to +8˚C Utilization: BCG, DPT, OPV, Measles, TT, Hep-B vaccine Holdover time: 24 hrs after 8 hrs continuous power supply Storage capacity: 300 ltr: 60,000 doses of mixed antigen & 20,000 doses of OPV 140 ltr : 25,000 doses of mixed antigen & 18,000 doses of OPV* * OPV and Measles for 1 month only. Store in DF if longer than a month
- 15. Right way of keeping vaccines in ILR• Keep all vaccines in baskets • Avoid placing vaccines at bottom of ILR. (never diluents, freeze sensitive) • Leave space between the vaccine boxes • Place a thermometer in the center of the ILR. • Same vaccines in same area. • Diluent / freeze sensitive/ Closer expiry date vaccines on top • Heat sensitive / Further expiry date vaccines in the bottom of basket
- 16. 4. Deep Freezer (DF) Size: 300 ltr / 140 ltr Level: District (300 ltr) / PHC (140 ltr) Temperature: -15o to -25o C Utilization: 1. Preparation of ice packs 2. Storing measles and OPV (only district) Holdover time: 24 hours with 8 hours supply Storage capacity: 300 ltr: 150, 000 to 200, 000 doses 140 ltr : Approx. 20 Ice Packs
- 17. Do’s for Deep freezer and ILR • Make one person responsible for loading, unloading, maintenance and temperature recording. • Install it in a cool and well ventilated room. • Place them at least 10-20 cm away from the wall. • Insure that it is LEVELLED. • Let the electrician fix the cable permanently inside the socket. • Use voltage stabilizer • Tape the switches in ON position so that there s no accidental switching off. • FIFO and EEFO
- 18. Contd… • Check the temperature at least every morning and evening. • Adjust the thermostat knob if the temperature is not correct. • Note the expiry date of all vaccines. • Clean and dry the chamber before loading. • Keep the lid locked. • Place the ILR and deep freezer in the same room.
- 19. Donts for deep freezer and ILR • Do not open the lid too often.. • Do not store other drugs • Don’t keep drinking water or food • Don’t keep more than one month supply • Don’t keep outdated Vac.s • Don’t fill ice packs to the top. • Don’t keep ILR and deep freezer hugging together.
- 21. The Cold Chain Room • ILRs and deep-freezers to be installed in a room that is – Not directly exposed to sunlight or any other source of heat. – Ventilated and protected from rain or flooding. • ILR and deep-freezers should be level, on wooden blocks, at least 10 cms away from wall • The plugs should be permanently fixed & labeled “DO NOT REMOVE” • Equipment should be locked and opened only if necessary • Keys to the equipment should be accessible
- 22. 5. Cold Boxes Size: 20 ltr and 5 ltr Level: District / PHC Temperature: +2˚C to +8˚C Utilization: All vaccines can be stored for transportation or in case of power failure Holdover time: 5 days (20 ltr) and 3 days (5 ltr) if unopened Storage capacity: 20 ltr: 52 Ice Packs & 6000 doses of mixed antigens 5 ltr: 20 Ice Packs & 1500 doses of mixed antigens
- 23. • Cold boxes. For transportation. • Fully frozen packs at the sides and bottom • Vac.s in polythene bags • DPT, DT, TT & diluents not to be kept in direct contact with frozen ice packs • Vaccine carriers: 4 frozen packs at the sides. • For small quantities
- 24. 6. Vaccine Carriers Size: 1.7 ltr Level: PHC/ Sub Centre Temperature: +2˚C to +8˚C Utilization: All vaccines can be carried in small quantity for vaccination sessions Holdover time: 12 hours Storage capacity: 4 Ice Packs &15- 20 vials of mixed antigens
- 25. 7. Ice Packs Size: 763 X 90 X 33 mm Ice capacity: 360 ml Weight: 80 gm Level: District / PHC/ Sub Centre Temperature: +2˚C to +8˚C Utilization: line the walls of vaccine carrier/cold box Time to Freeze: 48 hours in DF at - 20˚C
- 26. 8. Foam Pads Material: Soft Foam Thickness: 30 mm with at least 6 incisions Utilization: • temporary lid for unopened vaccines inside the carrier • surface to hold, protect and keep cool opened vaccine vials
- 27. Preparing icepacks for use: Filling and Freezing • Fill icepacks with water to mark • Fit the sealing plug and screw on the lid tightly • Hold each ice-pack upside down and squeeze it to make sure it does not leak. • Place the icepacks in the deep freezer. • Ice-packs need not be refilled every time they are used. The same water can be used repeatedly. • Do not use saline water for filling
- 28. Preparing icepacks for use: Conditioning • On the session day, take the frozen ice-packs you need from the freezer and place on a table • Allow ice-packs to sweat at room temperature for 15 minutes • Shake the ice pack to listen to melted for water. A Conditioned an ice-pack
- 31. Vaccines vulnerable to heat BCG (after reconstitution) OPV Measles DPT BCG (before reconstitution) DT TT Hep B
- 32. Vaccines vulnerable to Freezing Hep- B DPT DT TT
- 33. Vaccine Vial Monitor (VVM) The square is lighter than the circle. If the expiry date is not passed, use the vaccine The square is lighter than the circle. If the expiry date is not passed, use the vaccine The square matches the circle. Do not use the vaccine. Inform your supervisor The square is darker than the circle. Do not use the vaccine. Inform your supervisor
- 34. Summary of Vaccine Vulnerabilities Vaccine Heat Light Freezing Temperature at PHC OPV (live attenuated) Sensitive Sensitive Okay to freeze +2˚C to +8˚C (-15˚C to -25˚C at state, regional and district stores) BCG (live attenuated) Sensitive Sensitive Okay to freeze (before reconstitution) +2˚C to +8˚C Measles (live attenuated) Sensitive Sensitive Okay to freeze (before reconstitution) +2˚C to +8˚C (-15˚C to -25˚C at state, regional and district stores) DPT (toxoid, killed) Relatively heat stable Freezes at -3˚C. Discard if frozen. +2˚C to +8˚C Hep B (recombinant) Relatively heat stable Freezes at- .5˚C. Discard if frozen. +2˚C to +8˚C TT (toxoid) Relatively heat stable Freezes at -3˚C. Discard if frozen. +2˚C to +8˚C
- 35. HAZARDS OF IMMUNIZATION • No immune response is entirely free from the risk of adverse reactions or remote squeal. The adverse reactions that may occur may be grouped under the following heads: 1. Reactions inherent to inoculation 2. Reactions due to faulty techniques 3. Reactions due to hypersensitivity 4. Neurological involvement 5. Provocative reactions 6. Others
- 36. • 1. Reactions inherent to inoculation: These may be local general reactions. The local reactions may be pain, swelling, redness, tenderness and development of a small nodule or sterile abscess at the site of injection. • The general reactions may be fever, malaise, headache and other constitutional symptoms. Most killed bacterial vaccines (e.g., typhoid) cause some local and general reactions. Diphtheria and tetanus toxoids and live polio vaccine cause little reaction.
- 37. • 2. Reactions due to faulty techniques: Faulty techniques may relate to • faulty production of vaccine (e.g. inadequate inactivation of the microbe, inadequate detoxication), • too much vaccine given in one dose, • improper immunization site or route, • vaccine reconstituted with incorrect diluents, • wrong amount of diluent used, • drug substituted for vaccine or diluent, • vaccine prepared incorrectly for use (e.g., an adsorbed vaccine not shaken properly before use), • vaccine or dliluent contaminated, • vaccine stored incorrectly, • contraindications ignored (e.g. a child who experienced a severe reaction after a previous dose of DPT vaccine is immunized with he same vaccine), • reconstituted vaccine of one session of immunization used again at the subsequent session.
- 38. • Use of improperly sterilized syringes and needles carry the hazard of hepatitis B virus, and staphylo - and streptococcal infection
- 39. • 3. Reactions due to hypersensitivity: • Administration of antisera (e.g., ATS) may occasionally give rise to anaphylactic shock and serum sickness. Many viral vaccines contain traces of various antibiotics used in their preparation and some individuals may be sensitive to the antibiotic which it contains. Anaphylactic shock is a rare but dangerous complication of injection of antiserum. There is bronchospasm, dyspnoea, pallor, hypotension and collapse. • The symptoms may appear within a few minutes of injection or may be delayed up to 2 hours. Some viral vaccines prepared from embryonated eggs (e.g., influenza) may bring about generalized anaphylactic reactions. Serum sickness is characterized by symptoms such as fever, rash, oedema and joint pains occurring 7 -12 days of injection of antiserum.
- 40. • 4. Neurological involvement: • Neuritic manifestations may be seen after the administration of serum or vaccine. The well-known examples are the post-vaccinial encephalitis and encephalopathy following administration of anti -rabies and smallpox vaccines. • Guillain-Barre syndrome in association with the swine influenza vaccine is another example.
- 41. • 5. Provocative reactions: • Occasionally following immunization there may occur a disease totally unconnected with the immunizing agent (e.g., provocative polio after DPT or DT administration against diphtheria). • The mechanism seems to be that the individual is harboring the infectious agent and the administration of the vaccine shortens the incubation period and produces the disease or what may have been otherwise only a latent infection is converted into a clinical attack.
- 42. • 6. Others: • These may comprise damage to the fetus (e.g., with rubella vaccination); displacement in the age-distribution of a disease (e.g., a potential problem in mass vaccination against measles, rubella and mumps).
- 43. Irritability, malaise & systemic symptoms COMMON, MINOR REACTIONS Fever >38o C BCG Hib HepB Measles/ MMR Polio (OPV) DTP (pertussis) Tetanus 90-95% 5-15% Adults: 15%; Children: 5% ~10% - Up to 50% ~10%* - 2-10% - 5-15% <1% Up to 50% ~10% - - 1-6% 5% rash <1%** Up to 55% ~25% * Rate of local reactions likely to increase with booster doses, up to 50-85% ** Symptoms include diarrhoea, headache, and/or muscle pains Vaccine Local reaction (pain, swelling, redness)
- 44. RARE, MORE SERIOUS REACTIONS 0.76-1.3 (1st dose) 0.17 (subsequent doses) 0.15 (contacts) 4-30 daysVaccine-associated paralytic poliomyelitis (VAPP) Risk is higher for first dose, adults, and immunocompromised OPV 333 33 1-50 5-12 days 15-35 days 0-1 hour Febrile seizures Thrombocytopaenia Anaphylaxis Measles /MMR 1-2 5 0-1 hour 1-6 weeks Anaphylaxis Guillain Barré syndrome Hep B Nil knownHib 100-1000 1-700 2 2-6 months 1-12 months 1-12 months Suppurative lymphadenitis BCG osteitis Disseminated BCG BCG Rate per million doses Onset interval ReactionVaccine
- 45. RARE, MORE SERIOUS REACTIONS (2) 1000-60 000 570 570 20 0-1 0-24 hours 0-3 days 0-24 hours 0-1 hour 0-3 days Persistent (>3 hrs) inconsolable screaming Seizures Hypotonic, hyporesponsive episode (HHE) Anaphylaxis/shock Encephalopathy DPT Nil extra to tetanus reactionsTetanus-diphtheria 5-10 1-6 6-10 2-28 days 0-1 hour 1-6 weeks Brachial neuritis Anaphylaxis Sterile abscess Tetanus Rate per million doses Onset interval ReactionVaccine
- 46. RARE, MORE SERIOUS REACTIONS (3) 500-4000 in infants<6 months 5-20 7-21 days 0-1 hours Post-vaccination Encephalitis Allergic reaction/anaphylaxis Yellow fever 10-1000 1-2.3 Serious allergic reaction Neurological event Japanese encephalitis Rate per million doses Onset intervalReactionVaccine
Editor's Notes
- Common, minor reactions These occur within a day or two of immunization (except for measles/MMR - 6 to 12 days after immunization) and they only last one to a few days. Local reactions include pain, swelling and/or redness at the injection site and can be expected in about 10% of vaccinees, except for those injected with DTP, or tetanus boosters, where up to half can be affected. BCG causes a specific local reaction that starts as a papule (lump) two or more weeks after immunization that then becomes ulcerated and heals after several months, leaving a scar. Individuals with dormant tuberculosis infection often have an accelerated response to BCG. Keloid (thickened scar tissue) from the BCG lesion is more common among Asian and African populations. Systemic reactions include fever and occur in about 10% or less of vaccinees, except for DTP where it is again about half. Other common systemic reactions (e.g., irritability, malaise, ‘off-colour’, anorexia) can also occur after DTP. For measles/MMR and OPV the systemic reactions arise from vaccine virus infection. Measles’ vaccine causes fever, rash and/or conjunctivitis, and affects 5-15% of vaccinees. It is very mild compared to ‘wild’ measles, but for severely immunocompromised individuals, it can be severe, even fatal. Vaccine reactions for mumps (swollen parotid gland) and rubella (arthralgia and swollen lymph nodes) affect less than 1% of children. Rubella vaccine causes symptoms more often in adults, with 15% suffering from arthralgia. Systemic reactions from OPV affect less than 1% of vaccinees with diarrhoea, headache, and/or muscle pain.
- Rare, more serious reactions Most of the rare and more serious vaccine reactions (e.g., seizures, thrombocytopaenia, hypotonic-hyporesponsive episodes, persistent inconsolable screaming) do not lead to long term problems. Anaphylaxis, while potentially fatal, is treatable without leaving any long term effects. Although encephalopathy is included as a rare reaction to measles or DTP vaccine, it is not certain that this is in fact caused by these vaccines. Seizures are mostly febrile and risk depends on age, with much lower risk in infants under the age of 4 months or over the age of six years. Reactions to measles/MMR (except allergic and anaphylaxis) do not occur if already immune (~90% of those receiving a second dose).
- Rare, more serious reactions Most of the rare and more serious vaccine reactions (e.g., seizures, thrombocytopaenia, hypotonic-hyporesponsive episodes, persistent inconsolable screaming) do not lead to long term problems. Anaphylaxis, while potentially fatal, is treatable without leaving any long term effects. Although encephalopathy is included as a rare reaction to measles or DTP vaccine, it is not certain that this is in fact caused by these vaccines. Seizures are mostly febrile and risk depends on age, with much lower risk in infants under the age of 4 months or over the age of six years. Reactions to measles/MMR (except allergic and anaphylaxis) do not occur if already immune (~90% of those receiving a second dose).
- Rare, more serious reactions Most of the rare and more serious vaccine reactions (e.g., seizures, thrombocytopaenia, hypotonic-hyporesponsive episodes, persistent inconsolable screaming) do not lead to long term problems. Anaphylaxis, while potentially fatal, is treatable without leaving any long term effects. Although encephalopathy is included as a rare reaction to measles or DTP vaccine, it is not certain that this is in fact caused by these vaccines. Seizures are mostly febrile and risk depends on age, with much lower risk in infants under the age of 4 months or over the age of six years. Reactions to measles/MMR (except allergic and anaphylaxis) do not occur if already immune (~90% of those receiving a second dose).
