Fluorescent Minerals
•Download as PPTX, PDF•
0 likes•926 views
A presentation of some of the beautiful fluorescent minerals glowing under ultraviolet light.
Report
Share
Report
Share
Recommended
Recommended
How can minerals deposits be formed; GEOLOGICAL PROCESSES; Ore Fluids; Ore Forming Processes; Concentrating Processes; Magmatic mineral deposits; Residual mineral deposits ; Placer deposits; Sedimentary mineral deposits; Metamorhogenic mineral deposits; Hydrothermal mineral deposits ; Magmatic Deposits
Cumulate deposits: fractional crystallization processes can concentrate metals (Cr, Fe, PGE, Pt, Ni, Ti, Diamond ))
Pegmatites : late staged crystallization forms pegmatites and many residual elements are concentrated (Li, Ce, Be, Sn, U, Rare Earths (REE), Feldspar, Mica, Gems).
magmatic deposits; Mode of Formation of Magmatic Ores Deposits; Mode of Formation of Orthomagmatic Ores ; Fractional Crystallization (or Crystal fractionation ); Magmatic (or Liquid ) Immiscibility; Simple crystallization without concentration (Dissemination); Segregation of early formed crystals; (Layer Types); Injection of material concentrated elsewhere by differentiation Residual liquid segregation; Residual liquid injection; Immiscible liquid segregation; Immiscible-liquid-injection; Early magmatic deposit; Late magmatic deposit; Types of Magmatic Ore Deposits:Chromite; Fe-Ti (± V) oxides; Ni – Cu – Fe (± Pt) sulfides; Platinum Group Elements (PGEs); REE, and Zr in Carbonatites; Diamond in kimberlites.Topic 3 ore forming processes and magmatic mineral deposits

Topic 3 ore forming processes and magmatic mineral depositsGeology Department, Faculty of Science, Tanta University
More Related Content
What's hot
How can minerals deposits be formed; GEOLOGICAL PROCESSES; Ore Fluids; Ore Forming Processes; Concentrating Processes; Magmatic mineral deposits; Residual mineral deposits ; Placer deposits; Sedimentary mineral deposits; Metamorhogenic mineral deposits; Hydrothermal mineral deposits ; Magmatic Deposits
Cumulate deposits: fractional crystallization processes can concentrate metals (Cr, Fe, PGE, Pt, Ni, Ti, Diamond ))
Pegmatites : late staged crystallization forms pegmatites and many residual elements are concentrated (Li, Ce, Be, Sn, U, Rare Earths (REE), Feldspar, Mica, Gems).
magmatic deposits; Mode of Formation of Magmatic Ores Deposits; Mode of Formation of Orthomagmatic Ores ; Fractional Crystallization (or Crystal fractionation ); Magmatic (or Liquid ) Immiscibility; Simple crystallization without concentration (Dissemination); Segregation of early formed crystals; (Layer Types); Injection of material concentrated elsewhere by differentiation Residual liquid segregation; Residual liquid injection; Immiscible liquid segregation; Immiscible-liquid-injection; Early magmatic deposit; Late magmatic deposit; Types of Magmatic Ore Deposits:Chromite; Fe-Ti (± V) oxides; Ni – Cu – Fe (± Pt) sulfides; Platinum Group Elements (PGEs); REE, and Zr in Carbonatites; Diamond in kimberlites.Topic 3 ore forming processes and magmatic mineral deposits

Topic 3 ore forming processes and magmatic mineral depositsGeology Department, Faculty of Science, Tanta University
What's hot (20)
Topic 3 ore forming processes and magmatic mineral deposits

Topic 3 ore forming processes and magmatic mineral deposits
ENERGY CRITICAL ELEMENTS WITH A SPECIAL EMPHASIS ON REE EXPLORATION

ENERGY CRITICAL ELEMENTS WITH A SPECIAL EMPHASIS ON REE EXPLORATION
Similar to Fluorescent Minerals
Similar to Fluorescent Minerals (10)
Recently uploaded
Recently uploaded (20)
PLANT DISEASE MANAGEMENT PRINCIPLES AND ITS IMPORTANCE

PLANT DISEASE MANAGEMENT PRINCIPLES AND ITS IMPORTANCE
Continuum emission from within the plunging region of black hole discs

Continuum emission from within the plunging region of black hole discs
GBSN - Microbiology Lab 1 (Microbiology Lab Safety Procedures)

GBSN - Microbiology Lab 1 (Microbiology Lab Safety Procedures)
Plasmapheresis - Dr. E. Muralinath - Kalyan . C.pptx

Plasmapheresis - Dr. E. Muralinath - Kalyan . C.pptx
The Scientific names of some important families of Industrial plants .pdf

The Scientific names of some important families of Industrial plants .pdf
word2vec, node2vec, graph2vec, X2vec: Towards a Theory of Vector Embeddings o...

word2vec, node2vec, graph2vec, X2vec: Towards a Theory of Vector Embeddings o...
GBSN - Microbiology (Unit 7) Microbiology in Everyday Life

GBSN - Microbiology (Unit 7) Microbiology in Everyday Life
Detectability of Solar Panels as a Technosignature

Detectability of Solar Panels as a Technosignature
A Giant Impact Origin for the First Subduction on Earth

A Giant Impact Origin for the First Subduction on Earth
National Biodiversity protection initiatives and Convention on Biological Di...

National Biodiversity protection initiatives and Convention on Biological Di...
mixotrophy in cyanobacteria: a dual nutritional strategy

mixotrophy in cyanobacteria: a dual nutritional strategy
Ostiguy & Panizza & Moffitt (eds.) - Populism in Global Perspective. A Perfor...

Ostiguy & Panizza & Moffitt (eds.) - Populism in Global Perspective. A Perfor...
WASP-69b’s Escaping Envelope Is Confined to a Tail Extending at Least 7 Rp

WASP-69b’s Escaping Envelope Is Confined to a Tail Extending at Least 7 Rp
Isolation of AMF by wet sieving and decantation method pptx

Isolation of AMF by wet sieving and decantation method pptx
Fluorescent Minerals
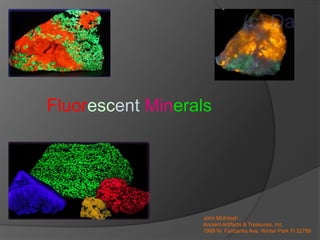
- 1. Fluorescent Minerals John McIntosh Ancient Artifacts & Treasures, Inc. 1999 W. Fairbanks Ave, Winter Park Fl 32789
- 2. Fluorescent Minerals What is a Fluorescent Mineral? These minerals have the ability to temporarily absorb a small amount of light and an instant later release a small amount of light of a different wavelength. This change in wavelength causes a temporary color change of the mineral in the eye of a human observer.
- 4. Fluorescent Minerals WHAT IS ULTRAVIOLET (UV)? Fluorescence is a "cold light" (luminescence) caused by electromagnetic radiation. There are many forms of electromagnetic radiation: visible light, radio, infrared, ultraviolet, X-rays, and gamma rays are all forms of electromagnetic radiation.
- 5. Fluorescent Minerals Ultraviolet radiation, as generated by a "black light" or Ultra Violet lights, is that portion of the electromagnetic spectrum that lies beyond the purple edge of the visible spectrum and has wavelengths between 100 and 400 nm. WHAT IS ULTRAVIOLET (UV)?
- 6. Fluorescent Minerals For the fluorescent mineral collector there are three useful wavelengths of ultraviolet light; Long wave, Mid wave, and Short wave. A few minerals fluoresce the same color in all wavelengths, others fluoresce in only one wavelength (usually SW), and yet others fluoresce different colors in different wavelengths. WHAT IS ULTRAVIOLET (UV)?
- 7. Fluorescent Minerals Fluorite: The Original "Fluorescent Mineral” One of the first people to observe fluorescence in minerals was George Gabriel Stokes in 1852. He noted the ability of fluorite to produce a blue glow when illuminated with invisible light "beyond the violet end of the spectrum." He called this phenomenon "fluorescence" after the mineral fluorite. The name has gained wide acceptance in mineralogy, gemology, biology, optics, commercial lighting and many other fields.
- 8. Fluorescent Minerals FLUORESCENCE IN MINERALS There are around 4,000 different types of minerals - approximately 15% of them are known to fluoresce. Impurities in the mineral (usually) cause this fluorescence - very few "pure" minerals are known to fluoresce.
- 9. Fluorescent Minerals Fluorescence usually occurs when specific impurities known as "activators" are present within the mineral. These activators are typically cations of metals such as: tungsten, molybdenum, lead, boron, titanium, manganese, uranium, and chromium. Rare earth elements such as europium, terbium, dysprosium, and yttrium are also known to contribute to the fluorescence phenomenon. Fluorescence can also be caused by crystal structural defects or organic impurities. Fluorescence “Activators
- 10. Fluorescent Minerals In addition to "activator" impurities, some impurities have a dampening effect on fluorescence. If iron or copper are present as impurities, they can reduce or eliminate fluorescence. Furthermore, if the activator mineral is present in large amounts, that can reduce the fluorescence effect Fluorescence “Dampeners”
- 11. Fluorescent Minerals Calcite Clam, Rucks Pit, FL
- 15. Fluorescent Minerals Raw and Faceted Diamonds
- 16. Fluorescent Minerals Blue Tourmaline (indicolite) in Quartz
- 25. Fluorescent Minerals https://www.facebook.com/groups/fluorescentminerals/ Fluorescent Mineral Groups
- 26. Fluorescent Minerals Fluorescent Mineral References [1] Basic Concepts in Fluorescence: Michael W. Davidson and others, Optical Microscopy Primer, Florida State University, last accessed October 2016. [2] Fluorescent Minerals: James O. Hamblen, a website about fluorescent minerals, Georgia Tech, 2003. [3] The World of Fluorescent Minerals, Stuart Schneider, Schiffer Publishing Ltd., 2006. [4] Dugway Geodes page on the SpiritRock Shop website, last accessed May 2017. [5] Collecting Fluorescent Minerals, Stuart Schneider, Schiffer Publishing Ltd., 2004. [6] Ultraviolet Light Safety: Connecticut High School Science Safety, Connecticut State Department of Education, last accessed October 2016. [7] A Contribution to Understanding the Effect of Blue Fluorescence on the Appearance of Diamonds: Thomas M. Moses and others, Gems and Gemology, Gemological Institute of America, Winter 1997. Good Reference Books