Polynuclear hydrocarbons module5
•
0 likes•70 views
Module 5- Naphthalene derivatives - naphthaquinones- 1-naphthaquinone- 2-naphthaquinone- amphi naphthaquinone- preparation-Reactions
Report
Share
Report
Share
Download to read offline
More Related Content
What's hot
What's hot (20)
Polynuclear Hydrocarbons Preparations and Reactions

Polynuclear Hydrocarbons Preparations and Reactions
POLYNUCLEAR HYDROCARBON : STRUCTURE AND USES OF NAPHTHELENE, ANTHRACENE,DI-PH...

POLYNUCLEAR HYDROCARBON : STRUCTURE AND USES OF NAPHTHELENE, ANTHRACENE,DI-PH...
Phenols: methods of preparation, chemical reaction

Phenols: methods of preparation, chemical reaction
Similar to Polynuclear hydrocarbons module5
Similar to Polynuclear hydrocarbons module5 (20)
Important name reaction for csir net and gate chemistry

Important name reaction for csir net and gate chemistry
Important name reaction for csir net and gate chemistry

Important name reaction for csir net and gate chemistry
naphthalene for organic chemistry to understand industrial chemistry

naphthalene for organic chemistry to understand industrial chemistry
Heterocyclic compounds - Thiophene - Synthesis of Thiophene - Characteristic ...

Heterocyclic compounds - Thiophene - Synthesis of Thiophene - Characteristic ...
More from syed mohamed
More from syed mohamed (9)
Recently uploaded
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Recently uploaded (20)
Call Girls Ahmedabad +917728919243 call me Independent Escort Service

Call Girls Ahmedabad +917728919243 call me Independent Escort Service
Use of mutants in understanding seedling development.pptx

Use of mutants in understanding seedling development.pptx
Porella : features, morphology, anatomy, reproduction etc.

Porella : features, morphology, anatomy, reproduction etc.
Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds

Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds
PATNA CALL GIRLS 8617370543 LOW PRICE ESCORT SERVICE

PATNA CALL GIRLS 8617370543 LOW PRICE ESCORT SERVICE
Genetics and epigenetics of ADHD and comorbid conditions

Genetics and epigenetics of ADHD and comorbid conditions
Bhiwandi Bhiwandi ❤CALL GIRL 7870993772 ❤CALL GIRLS ESCORT SERVICE In Bhiwan...

Bhiwandi Bhiwandi ❤CALL GIRL 7870993772 ❤CALL GIRLS ESCORT SERVICE In Bhiwan...
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
THE ROLE OF BIOTECHNOLOGY IN THE ECONOMIC UPLIFT.pptx

THE ROLE OF BIOTECHNOLOGY IN THE ECONOMIC UPLIFT.pptx
Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b

Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b
Polynuclear hydrocarbons module5
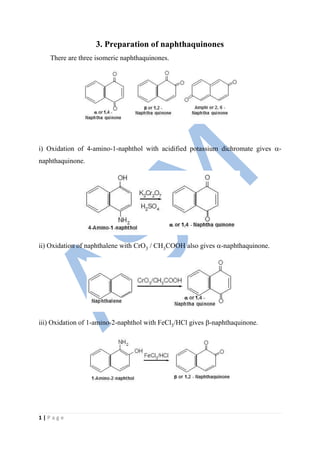
- 1. 1 | P a g e 3. Preparation of naphthaquinones There are three isomeric naphthaquinones. i) Oxidation of 4-amino-1-naphthol with acidified potassium dichromate gives - naphthaquinone. ii) Oxidation of naphthalene with CrO3 / CH3COOH also gives -naphthaquinone. iii) Oxidation of 1-amino-2-naphthol with FeCl3/HCl gives -naphthaquinone.
- 2. 2 | P a g e iv) Oxidation of 2, 6 - dihydroxy naphthalene with PbO2 gives amphi naphthaquinone. -Naphthaquinone is also prepared by the oxidation of 1,4-Dihydroxy naphthalene and naphthalene as given below Reactions of naphthaquinone i) -Naphthaquinone is reduced by zinc and HCl to 1,4-dihydroxynaphthalene. On oxidation it gives phthalic acid. ii) It forms a monoxime which exhibits tautomerism.
- 3. 3 | P a g e iii) On reaction with nitrous acid it is converted to 1,3-diketohydrindene. Prepared by Dr. A. Syed Mohamed HoD and Senior Assistant Professor, Research Dept. of Chemistry Sadakathullah Appa College (Autonomous) Tirunelveli, Tamilnadu, India. asm2032@gmail.com The author ackowledges Prof. H. Kassali Rahmathullah, Dr. T. Syed Ismail, Dr. M. Kamalutheen, Former Professor, Dept. of Chemistry, Sadakathullah Appa College for their valuable contribution.
