Polynuclear hydrocarbons Module4
•
0 likes•46 views
Module 4 - Naphthalene derivatives - Naphthyl amines- 1-Naphthyl amines - 2- Naphthyl amines - Preparation - Bucherer reaction- Reactions of naphthylamines
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
What's hot
What's hot (20)
Important name reaction for csir net and gate chemistry

Important name reaction for csir net and gate chemistry
21.1 - Part 1 Structure and Properties of Carboxylic Acid Derivatives - Wade 7th

21.1 - Part 1 Structure and Properties of Carboxylic Acid Derivatives - Wade 7th
General Chemistry 2 Assignment - Preparation of amine (Group 13 and 18)

General Chemistry 2 Assignment - Preparation of amine (Group 13 and 18)
Similar to Polynuclear hydrocarbons Module4
Similar to Polynuclear hydrocarbons Module4 (11)
Polynuclear Hydrocarbons Preparations and Reactions

Polynuclear Hydrocarbons Preparations and Reactions
naphthalene for organic chemistry to understand industrial chemistry

naphthalene for organic chemistry to understand industrial chemistry
Molecular rearrangements involving electron deficient nitrogen as an intermed...

Molecular rearrangements involving electron deficient nitrogen as an intermed...
humnamehmood3219chm-627pinacolpinacolonerearrangementppt-210225074927-1.pdf

humnamehmood3219chm-627pinacolpinacolonerearrangementppt-210225074927-1.pdf
Heterocyclic compounds containing mono hetero atom pptx

Heterocyclic compounds containing mono hetero atom pptx
More from syed mohamed
More from syed mohamed (12)
Recently uploaded
Ultrasound color Doppler imaging has been routinely used for the diagnosis of cardiovascular diseases, enabling real-time flow visualization through the Doppler effect. Yet, its inability to provide true flow velocity vectors due to its one-dimensional detection limits its efficacy. To overcome this limitation, various VFI schemes, including multi-angle beams, speckle tracking, and transverse oscillation, have been explored, with some already available commercially. However, many of these methods still rely on autocorrelation, which poses inherent issues such as underestimation, aliasing, and the need for large ensemble sizes. Conversely, speckle-tracking-based VFI enables lateral velocity estimation but suffers from significantly lower accuracy compared to axial velocity measurements.
To address these challenges, we have presented a speckle-tracking-based VFI approach utilizing multi-angle ultrafast plane wave imaging. Our approach involves estimating axial velocity components projected onto individual steered plane waves, which are then combined to derive the velocity vector. Additionally, we've introduced a VFI visualization technique with high spatial and temporal resolutions capable of tracking flow particle trajectories.
Simulation and flow phantom experiments demonstrate that the proposed VFI method outperforms both speckle-tracking-based VFI and autocorrelation VFI counterparts by at least a factor of three. Furthermore, in vivo measurements on carotid arteries using the Prodigy ultrasound scanner demonstrate the effectiveness of our approach compared to existing methods, providing a more robust imaging tool for hemodynamic studies.
Learning objectives:
- Understand fundamental limitations of color Doppler imaging.
- Understand principles behind advanced vector flow imaging techniques.
- Familiarize with the ultrasound speckle tracking technique and its implications in flow imaging.
- Explore experiments conducted using multi-angle plane wave ultrafast imaging, specifically utilizing the pulse-sequence mode on a 128-channel ultrasound research platform. (May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...

(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...Scintica Instrumentation
+971581248768 Mtp-Kit (500MG) Prices » Dubai [(+971581248768**)] Abortion Pills For Sale In Dubai, UAE, Mifepristone and Misoprostol Tablets Available In Dubai, UAE CONTACT DR.Maya Whatsapp +971581248768 We Have Abortion Pills / Cytotec Tablets /Mifegest Kit Available in Dubai, Sharjah, Abudhabi, Ajman, Alain, Fujairah, Ras Al Khaimah, Umm Al Quwain, UAE, Buy cytotec in Dubai +971581248768''''Abortion Pills near me DUBAI | ABU DHABI|UAE. Price of Misoprostol, Cytotec” +971581248768' Dr.DEEM ''BUY ABORTION PILLS MIFEGEST KIT, MISOPROTONE, CYTOTEC PILLS IN DUBAI, ABU DHABI,UAE'' Contact me now via What's App…… abortion Pills Cytotec also available Oman Qatar Doha Saudi Arabia Bahrain Above all, Cytotec Abortion Pills are Available In Dubai / UAE, you will be very happy to do abortion in Dubai we are providing cytotec 200mg abortion pill in Dubai, UAE. Medication abortion offers an alternative to Surgical Abortion for women in the early weeks of pregnancy. We only offer abortion pills from 1 week-6 Months. We then advise you to use surgery if its beyond 6 months. Our Abu Dhabi, Ajman, Al Ain, Dubai, Fujairah, Ras Al Khaimah (RAK), Sharjah, Umm Al Quwain (UAQ) United Arab Emirates Abortion Clinic provides the safest and most advanced techniques for providing non-surgical, medical and surgical abortion methods for early through late second trimester, including the Abortion By Pill Procedure (RU 486, Mifeprex, Mifepristone, early options French Abortion Pill), Tamoxifen, Methotrexate and Cytotec (Misoprostol). The Abu Dhabi, United Arab Emirates Abortion Clinic performs Same Day Abortion Procedure using medications that are taken on the first day of the office visit and will cause the abortion to occur generally within 4 to 6 hours (as early as 30 minutes) for patients who are 3 to 12 weeks pregnant. When Mifepristone and Misoprostol are used, 50% of patients complete in 4 to 6 hours; 75% to 80% in 12 hours; and 90% in 24 hours. We use a regimen that allows for completion without the need for surgery 99% of the time. All advanced second trimester and late term pregnancies at our Tampa clinic (17 to 24 weeks or greater) can be completed within 24 hours or less 99% of the time without the need surgery. The procedure is completed with minimal to no complications. Our Women's Health Center located in Abu Dhabi, United Arab Emirates, uses the latest medications for medical abortions (RU-486, Mifeprex, Mifegyne, Mifepristone, early options French abortion pill), Methotrexate and Cytotec (Misoprostol). The safety standards of our Abu Dhabi, United Arab Emirates Abortion Doctors remain unparalleled. They consistently maintain the lowest complication rates throughout the nation. Our Physicians and staff are always available to answer questions and care for women in one of the most difficult times in their lives. The decision to have an abortion at the Abortion Clinic in Abu Dhabi, (United Arab Emirates)+971581248768+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Recently uploaded (20)
300003-World Science Day For Peace And Development.pptx

300003-World Science Day For Peace And Development.pptx
Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds

Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds
(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...

(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Selaginella: features, morphology ,anatomy and reproduction.

Selaginella: features, morphology ,anatomy and reproduction.
Use of mutants in understanding seedling development.pptx

Use of mutants in understanding seedling development.pptx
Locating and isolating a gene, FISH, GISH, Chromosome walking and jumping, te...

Locating and isolating a gene, FISH, GISH, Chromosome walking and jumping, te...
development of diagnostic enzyme assay to detect leuser virus

development of diagnostic enzyme assay to detect leuser virus
FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry

FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry
Pulmonary drug delivery system M.pharm -2nd sem P'ceutics

Pulmonary drug delivery system M.pharm -2nd sem P'ceutics
Module for Grade 9 for Asynchronous/Distance learning

Module for Grade 9 for Asynchronous/Distance learning
Polynuclear hydrocarbons Module4
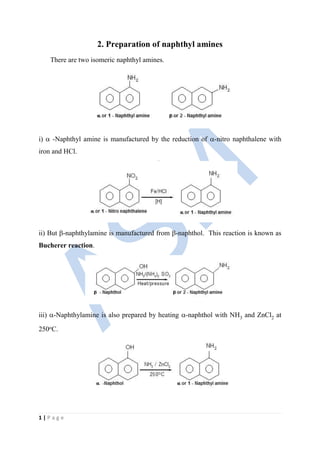
- 1. 1 | P a g e 2. Preparation of naphthyl amines There are two isomeric naphthyl amines. i) -Naphthyl amine is manufactured by the reduction of -nitro naphthalene with iron and HCl. ii) But -naphthylamine is manufactured from -naphthol. This reaction is known as Bucherer reaction. iii) -Naphthylamine is also prepared by heating -naphthol with NH3 and ZnCl2 at 250oC.
- 2. 2 | P a g e Reactions of naphthylamines i) Electrophilic substitution reactions: They undergo halogenation, nitration, sulphonation etc. However, to avoid formation of polysubstituted derivatives amino group must be protected through acylation. In case of -naphthylamine the incoming substituent generally enters at 4-position while in -naphthylamine it enters at 1-position. ii) Reduction : They are reduced by sodium and isoamyl alcohol. -Naphthylamine forming (5,6,7,8-tetrahydro-naphthylaine), while -naphthylamine gives 1,2,3,4- tetrahydro-2-naphthylaine. iii) Oxidation: They are highly susceptible to oxidation and reduce ammoniacal silver nitrate. Oxidation with acidified KMnO4 gives phthalic acid.
- 3. 3 | P a g e Prepared by Dr. A. Syed Mohamed HoD and Senior Assistant Professor, Research Dept. of Chemistry Sadakathullah Appa College (Autonomous) Tirunelveli, Tamilnadu, India. asm2032@gmail.com The author ackowledges Prof. H. Kassali Rahmathullah, Dr. T. Syed Ismail, Dr. M. Kamalutheen, Former Professor, Dept. of Chemistry, Sadakathullah Appa College for their valuable contribution.
