Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Climbing film evaporator || Pharmaceutical Engineering || Anurag Gupta

Climbing film evaporator || Pharmaceutical Engineering || Anurag Gupta
Study of principle, procedure, merits, demerits and applications of physical,...

Study of principle, procedure, merits, demerits and applications of physical,...
Similar to FREEZE DRYER (1).pptx
Similar to FREEZE DRYER (1).pptx (20)
Freeze-drying on Food Preservation technique by low temperature.pptx

Freeze-drying on Food Preservation technique by low temperature.pptx
More from sruthi's Pharma World
More from sruthi's Pharma World (18)
Materials of pharmaceutical plant construction.pptx

Materials of pharmaceutical plant construction.pptx
CENTRIFUGATION EQUIPMENT, PERFORATED BASKET CENTRIFUGE

CENTRIFUGATION EQUIPMENT, PERFORATED BASKET CENTRIFUGE
Recently uploaded
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Mehran University Newsletter is a Quarterly Publication from Public Relations OfficeMehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
https://app.box.com/s/x7vf0j7xaxl2hlczxm3ny497y4yto33i80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...Nguyen Thanh Tu Collection
Recently uploaded (20)
ICT Role in 21st Century Education & its Challenges.pptx

ICT Role in 21st Century Education & its Challenges.pptx
Plant propagation: Sexual and Asexual propapagation.pptx

Plant propagation: Sexual and Asexual propapagation.pptx
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
21st_Century_Skills_Framework_Final_Presentation_2.pptx

21st_Century_Skills_Framework_Final_Presentation_2.pptx
80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx

Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Food safety_Challenges food safety laboratories_.pdf

Food safety_Challenges food safety laboratories_.pdf
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx

HMCS Vancouver Pre-Deployment Brief - May 2024 (Web Version).pptx
FREEZE DRYER (1).pptx
- 1. FREEZE DRYER Sruthi’s Pharma World
- 2. OBJECTIVES • To preserve the biological activity of a product • To reduce the product weight to lower the transportation cost • To extend the shelf life or stability • To dry thermo labile materials • To eliminate the need for refrigerated storage • To get accurate sterile dosing in to the final product container Sruthi’s Pharma World
- 3. PRINCIPLE • Freeze drying is also known as lyophilization • lyophilisation is carried out using a simple principle of physics, sublimation • Sublimation is the transition of substance from the solid to the vapor state without passing through an intermediate liquid phase. Sruthi’s Pharma World
- 4. PRINCIPLE • The entire process is performed at a low temperature and pressure (usually below the triple point) by applying vacuum, hence it is suited for drying of thermolabile compounds Sruthi’s Pharma World
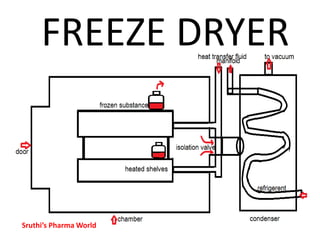
- 5. CONSTRUCTION • A drying chamber in which trays are loaded • Heat source : radiation source, heating coils • Vapor condensing or adsorption system • Vacuum pump or steam ejector or both Sruthi’s Pharma World
- 6. CONSTRUCTION • The distance between subliming surface and condenser must be less than the mean path of the molecules • This increases the rate of drying Sruthi’s Pharma World
- 7. CONSTRUCTION • The temperature of the condenser must be much lower than the evaporated surface of frozen substance (to maintain this condition condenser surface must be cleaned repeatedly) Sruthi’s Pharma World
- 8. WORKING 1. Preparation And Pretreatment 2.Prefreezing For Solidifying Water 3.Primary Drying(sublimation Of Ice Under Vacuum) 4.Secondary Drying(removal Of Residual Moisture Under High Vacuum) 5.Packing Sruthi’s Pharma World
- 9. PREPARATION AND PRETREATMENT • The solution is pre-concentrated under normal vacuum tray drying • This reduces the actual drying by 8-10 times Sruthi’s Pharma World
- 10. PRE-FREEZING TO SOLIDIFY WATER • The sample should be packed in vials, ampoules, or bottles • Kept in cold shelves (about-50°c) • During this stage cabinet is maintained at low temperature and pressure Sruthi’s Pharma World
- 11. PRIMARY DRYING • After freezing, the product is placed under vacuum. • Heat is applied to frozen product to accelerate sublimation • The temperature and pressure should be below the triple point of water i.e., 0.0098°c and 0.533kilopascals for sublimation when water alone is present Sruthi’s Pharma World
- 12. PRIMARY DRYING • Vacuum is applied to the tune of 3mmhg • The temperature is increased to about 30°c in a span of 2 hours • The driving force for sublimation: the temperature and vapor pressure difference between evaporating surface and condenser Sruthi’s Pharma World
- 13. PRIMARY DRYING • This enables the frozen solvent in the product to vaporize without passing through liquid phase, a process known as sublimation • Easily removes moisture up to 98% to 99% Sruthi’s Pharma World
- 14. SECONDARY DRYING • Removal of residual moisture under high vacuum . • The temperature is raised to 50°c-60°c and vacuum is lowered about 50mmhg • Bound water is removed • Rate of drying is slow • It takes about 10-20 hrs Sruthi’s Pharma World
- 15. PACKING • After vacuum is replaced by inert gas, the bottles and vials are closed Sruthi’s Pharma World
- 16. CHARACTERISTICS OF FREEZE DRIED PRODUCT • Sufficient strength • Uniform color • Sufficiently dry • Sufficiently porous • Sterile • Free of pyrogens and particulates • Chemically stable both in dry and reconstitution Sruthi’s Pharma World
- 17. ADVANTAGES Removal of water at low temperature Thermo labile materials can be dried Compatible with aseptic operations Sterility can be maintained Reconstitution is easy Loss of volatile material is less Sruthi’s Pharma World
- 18. DISADVANTAGES The product is prone to oxidation due to high porosity and large surface area Cost may be an issue depending on the product Long time process Sruthi’s Pharma World
- 19. USES • Used for drying blood plasma and fractionated products bacterial and viral cultures human tissues Sruthi’s Pharma World
- 20. THANK YOU
