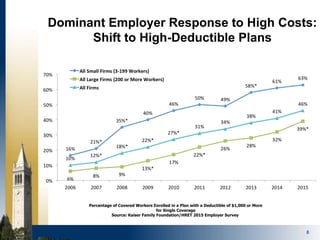
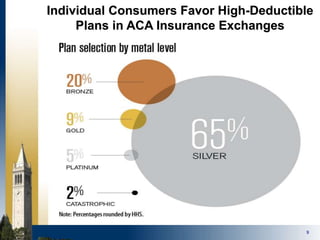
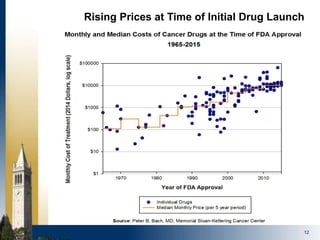
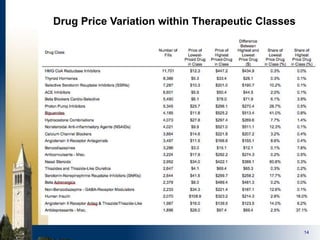
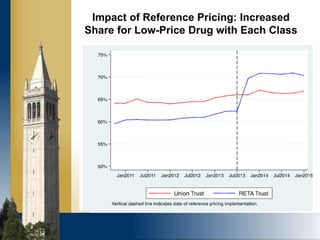
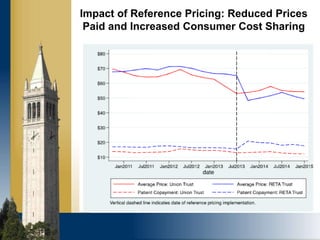
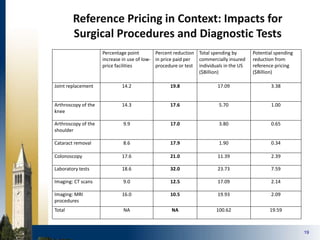
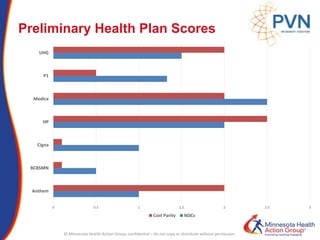
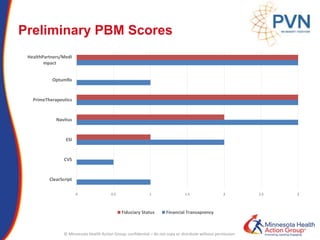
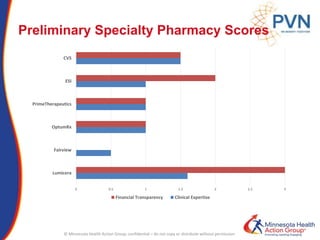
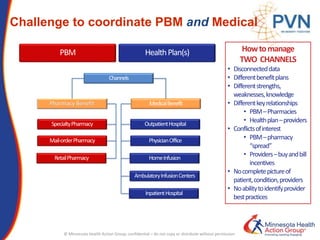
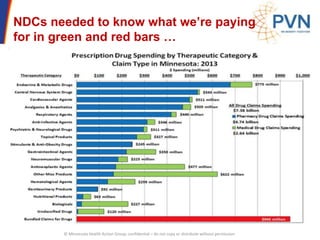
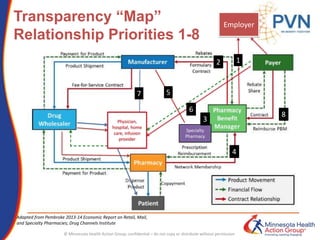
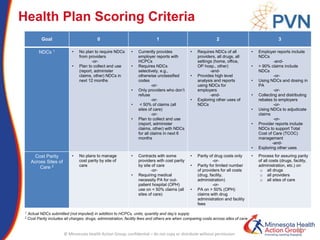
The document outlines strategies for purchasers dealing with high pharmaceutical prices, focusing on reference pricing as a method to manage costs. It highlights the importance of transparency and innovative employer strategies in managing specialty drugs and discusses the challenges faced by purchasers in implementing these strategies. Additionally, it emphasizes the need for better consumer information and collaboration among stakeholders to optimize healthcare spending.