The document discusses various topics related to understanding patents, including:
- What a patent is and the different types of patents including utility patents, design patents, and plant patents.
- The types of patent applications including provisional, regular, PCT, and foreign applications.
- Key patent terms and dates such as priority date, filing date, publication date, grant/issue date, and expiry date.
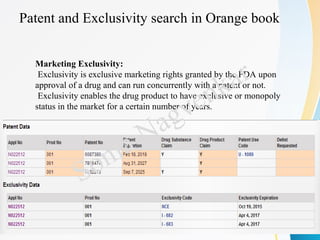
- How to read and understand a patent including terminology, claims, references, and kind codes.
- What constitutes patent infringement and how independent and dependent claims relate to determining infringement.