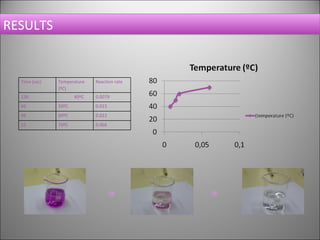
This document summarizes an experiment that investigates the effect of temperature on reaction rate. The experiment uses the reaction between potassium permanganate and oxalic acid, where the disappearance of the purple permanganate color indicates the extent of reaction. The experiment is conducted at different temperatures (40, 50, 60, 70°C), with all other variables kept constant. The results show that increasing the temperature leads to faster reaction rates, as indicated by shorter reaction times.