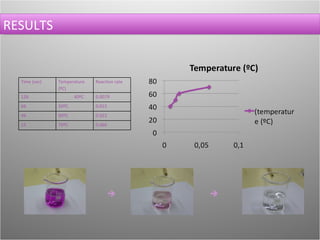
This document describes an experiment to observe the effect of temperature changes on reaction rate. The experiment involves a reaction between potassium permanganate, sulfuric acid, and oxalic acid. The reaction rate is determined by measuring the time it takes for the purple permanganate solution to become colorless. Results show that as temperature increases from 40°C to 70°C, the reaction time decreases and the reaction rate increases. The conclusion is that higher temperatures lead to faster reaction rates.