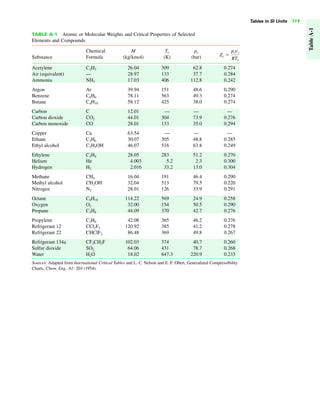
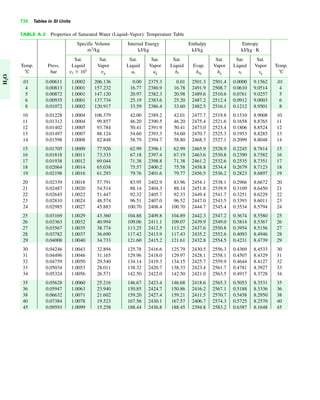
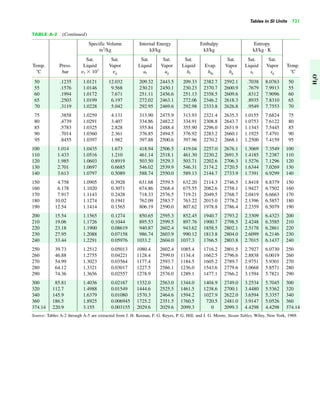
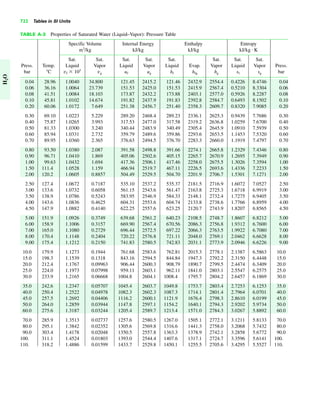
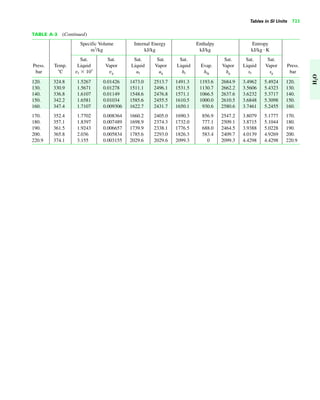
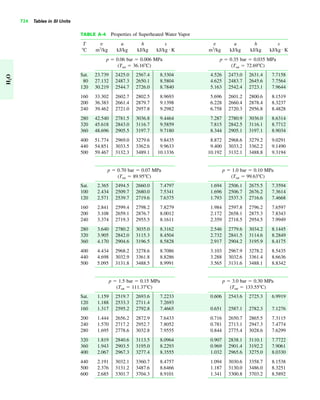
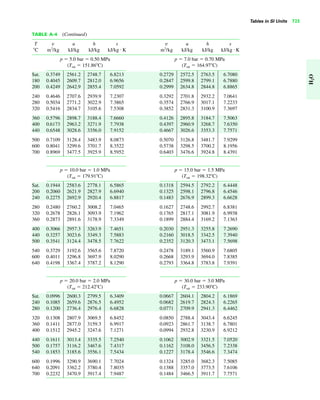
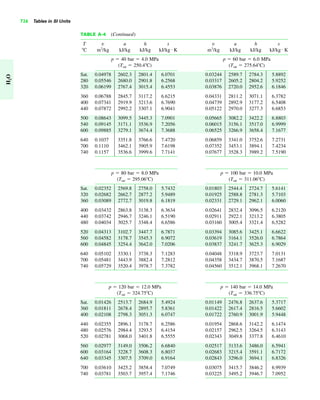
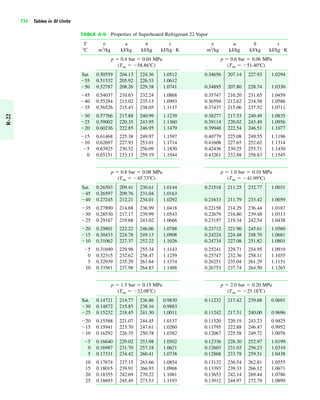
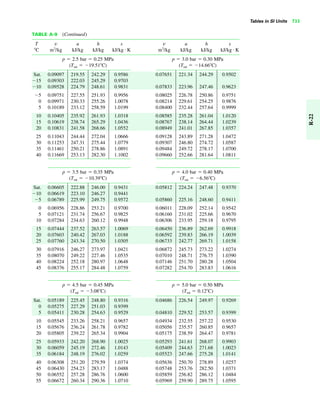
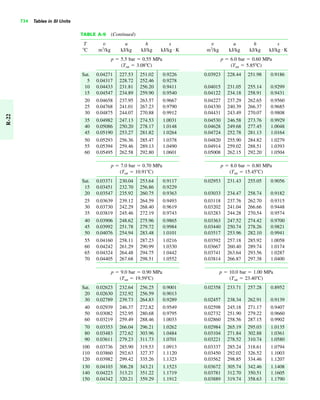
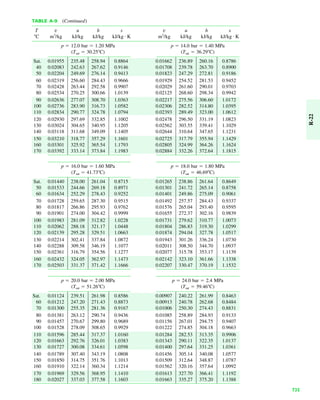
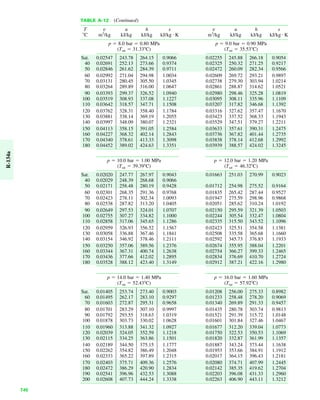
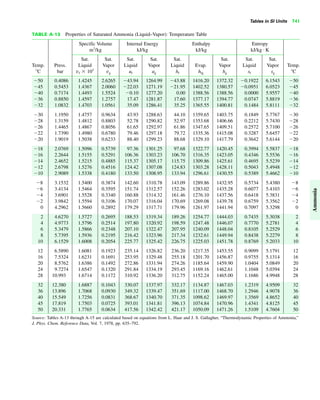
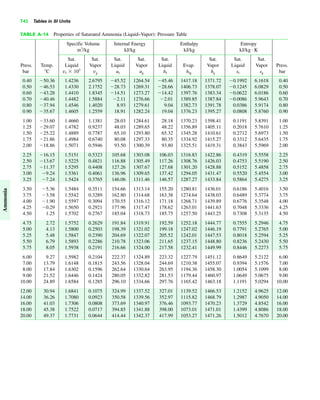
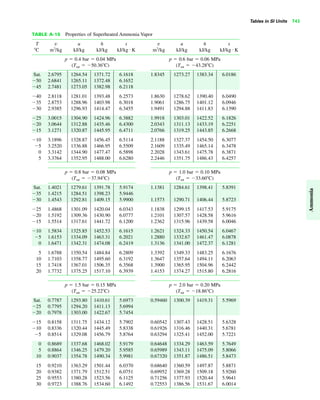
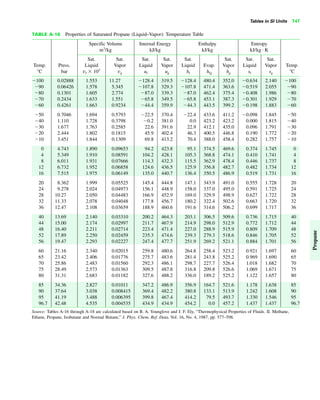
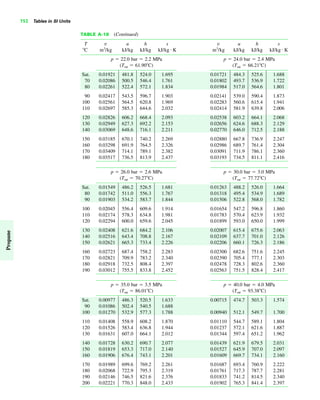
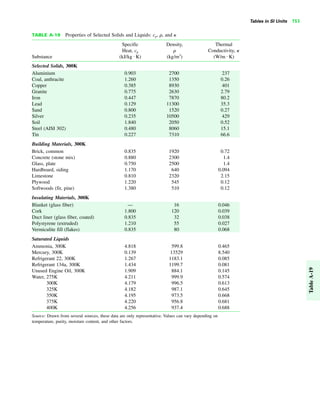
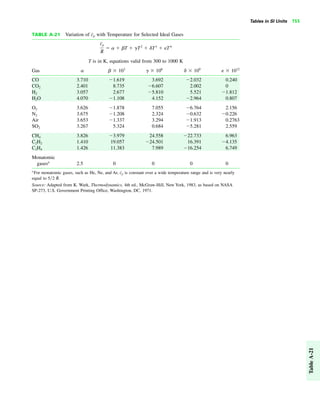
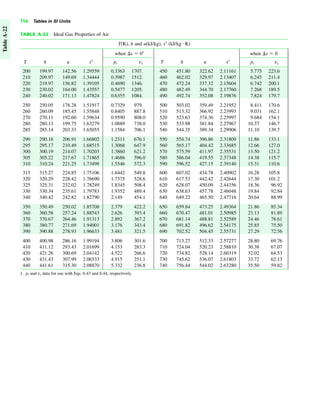
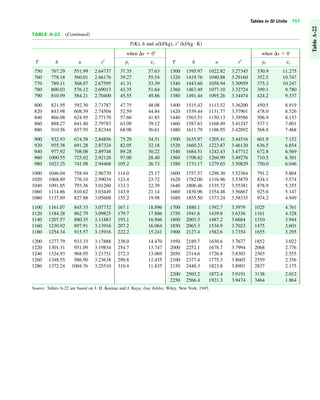
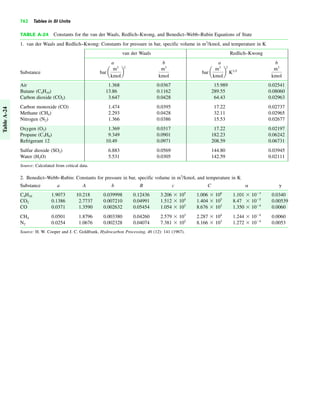
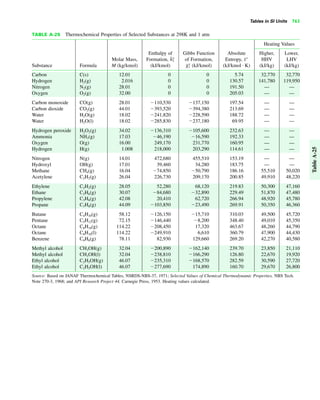
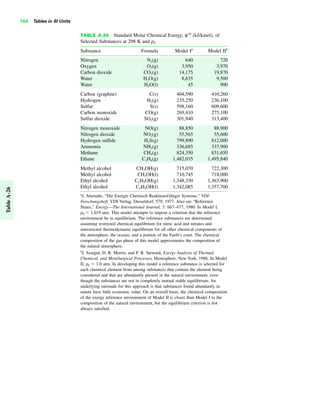
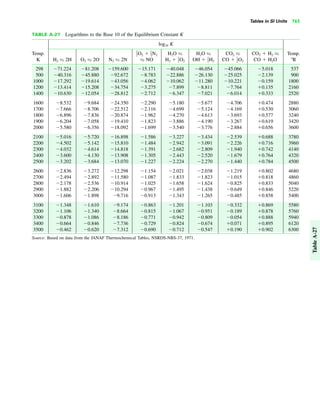
The document contains a series of tables detailing the atomic or molecular weights and critical properties of selected elements and compounds, including various refrigerants and water properties in different phases. It lists important thermodynamic properties such as temperature, pressure, specific volume, internal energy, enthalpy, and entropy for these substances. Additionally, it provides ideal gas properties and constants for various equations of state.