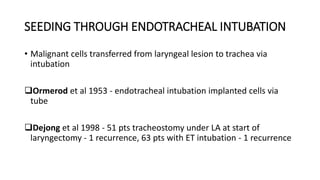
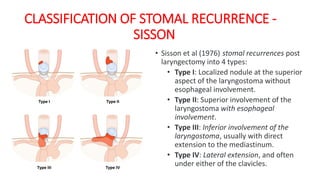
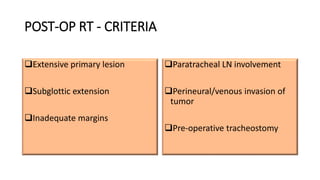
Stomal recurrence after laryngectomy occurs in 1.7-15% of patients and is usually fatal. Risk factors include subglottic tumor location, advanced T and N stage, pre-operative tracheostomy, and positive margins. Surgery is the main treatment for localized recurrence, involving wide excision and mediastinal lymph node dissection. Post-operative radiation may prevent recurrence in high risk patients by sterilizing the area. Close follow-up is important to detect recurrence early.