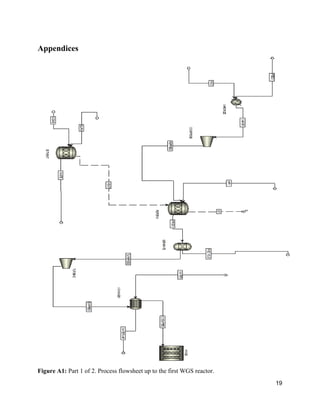
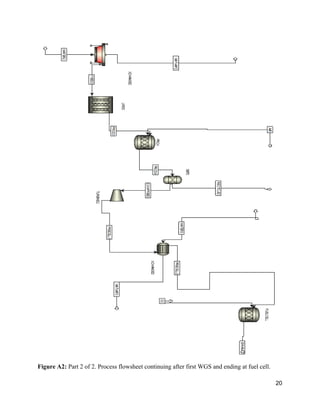
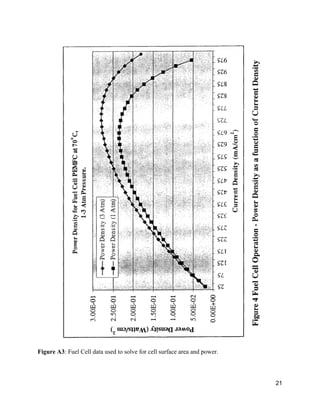
The document describes a process design for a proton exchange membrane (PEM) fuel cell power plant that produces hydrogen from heptane to deliver electricity to a 300 office building. Simulations were run using ASPEN software to model and optimize the plant design, which includes an auto-reformer, water-gas shift reactors, a preferential oxidation reactor, and heat exchangers. An economic analysis was conducted to determine the total capital costs and profitability of the plant over 25 years. The current plant design is not able to fully power the processing needs and falls short of the electrical demand, requiring further optimization to improve efficiency and meet all power requirements.





− rco = η − β
(9)n k n k /(RT)l = l o − E
where is the effectiveness factor accounting for intraparticle mass transport limitation, [CO] is η
the gas phase concentration of CO, , is the equilibrium CO ][H ]/{[H O][CO]K }β = [ 2 2 2 T KT
constant for the WGS reaction, and .n(K ) 577.8/T .33l T = 4 − 4
The PROX (PFR) reactor is a packed catalyst bed where the main and side reactions are,
CO O₂ CO₂ (main reaction)2 + = 2 (10)
H₂ O₂ H₂ (side reaction)2 + = 2 (11)
The PROX reactor kinetics model is shown in the equation,
(12)1 /Q ) XCO = 1 − ( − η * k1 * k2 Total
1.66
(13).58 exp(− 522/T[K])k1 = 7 * 106
8
(14)6.2 y k2 = 2 * P0.4
CO,in
−0.6
* λ0.82
* mPt
where XCO is fractional conversion, is the effectiveness factor of 0.5, k1k2=12 std cm3
/min, P isη
the total pressure, yCO,in is the carbon monoxide mole fraction in the feed to PROX,
, mPt is the mass of Pt in the catalyst, and QTotal is the std cm3
/min of feed to theO ]/[CO]λ = 2 * [ 2
PROX.
Heat Exchangers, turbines, compressors, and separators were used throughout our
simulation to cool the feed, expand the pressure in the feed, and compress or separate the gases
5](https://image.slidesharecdn.com/3bc49630-39e0-4ec5-b64e-6f6bafb24d73-150421023631-conversion-gate01/85/Stationary-Fuel-Cell-Lab-Report-6-320.jpg)












![inlet cooling streams which would reduce the total amount of water used. The design could also be
optimized by utilizing recycle streams, although not modeled in the simulation. Further optimization of
this recycle design could reduce the total amount of heptane feed required in the plant, and optimize the
plant is such a way that the heat exchangers primarily use the water produced from the fuel cell, and
only use external water if necessary.
References
[1] Weider, John W., et al. “Engineering a Membrane Electrode Assembly” The Electrochemical
Society Interface (2003): 4143. Print.
[2] Energy.gov. U.S. Department of Energy, Hydrogen and Fuel Cell Technology Basics, 2013.
Web. 10 Mar. 2015.
[3] U.S. Energy Information Administration, Form EIA826, Monthly Electric Sales and
Revenue Report with State Distributions Report
[4] ARAMARK Energy Services, “Best Practices in Energy Procurement” Managed Heat Rate
[5] FuelCellToday, The Fuel Cell Industry Review 2013, Johnson Matthey PLC trading
[6] Kamarudin, S.K.“Technical design and economic evaluation of a PEM fuel cell system”
ScienceDirect 157.2 (2006): 641–649. Print.
18](https://image.slidesharecdn.com/3bc49630-39e0-4ec5-b64e-6f6bafb24d73-150421023631-conversion-gate01/85/Stationary-Fuel-Cell-Lab-Report-19-320.jpg)