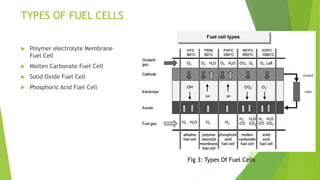
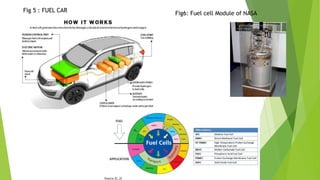
The document is a technical seminar report on superconductors submitted by a student. It discusses different types of fuel cells including polymer electrolyte membrane fuel cells, molten carbonate fuel cells, solid oxide fuel cells, and phosphoric acid fuel cells. It describes the major components of a fuel cell and their applications in areas like transportation, space, and portable electronics. The advantages of fuel cells are also highlighted such as high energy efficiency and low emissions, while the disadvantages include high costs and lack of widespread availability.