Slider post_PR-07_14 Sep 1.pdf
•
0 likes•5 views
Benefit-Risk Determination is defined as “the analysis of all assessments of benefit and risk of possible relevance for the use of the device for the intended purpose, when used in accordance with the intended purpose given by the manufacturer
Report
Share
Report
Share
Download to read offline
Recommended
Application of Quality Risk Management in Commissioning & Qualifcation

Application of Quality Risk Management in Commissioning & QualifcationGMP EDUCATION : Not for Profit Organization
PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...

PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...GAFM Academy of Project Management ® - ISO 29990 Certified International Board of Standards
More Related Content
Similar to Slider post_PR-07_14 Sep 1.pdf
Application of Quality Risk Management in Commissioning & Qualifcation

Application of Quality Risk Management in Commissioning & QualifcationGMP EDUCATION : Not for Profit Organization
PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...

PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...GAFM Academy of Project Management ® - ISO 29990 Certified International Board of Standards
Similar to Slider post_PR-07_14 Sep 1.pdf (20)
Accelerating the Development of Medical Devices: The Value of Proactive Risk ...

Accelerating the Development of Medical Devices: The Value of Proactive Risk ...
Ich guidelines on risk assessment and risk mangment

Ich guidelines on risk assessment and risk mangment
ISO: 14971 Quality risk management of medical devices

ISO: 14971 Quality risk management of medical devices
Risk-Management-Risk-Assessment-Risk-Mitigation-Risk-Monitoring.pptx

Risk-Management-Risk-Assessment-Risk-Mitigation-Risk-Monitoring.pptx
Toward a Trusted Supply Chain White Paper from Microsoft

Toward a Trusted Supply Chain White Paper from Microsoft
Final Class Presentation on Determining Project Stakeholders & Risks.pptx

Final Class Presentation on Determining Project Stakeholders & Risks.pptx
Application of Quality Risk Management in Commissioning & Qualifcation

Application of Quality Risk Management in Commissioning & Qualifcation
AbstractKey FeaturesAssessmentIntroductionMeasur.docx

AbstractKey FeaturesAssessmentIntroductionMeasur.docx
PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...

PROJECT RISK MANAGEMENT ... complete training materials and others at www.ga...
Practical Application Of System Safety For Performance Improvement

Practical Application Of System Safety For Performance Improvement
Session 04_Risk Assessment Program for YSP_Risk Analysis I

Session 04_Risk Assessment Program for YSP_Risk Analysis I
Step by-step for risk analysis and management-yaser aljohani

Step by-step for risk analysis and management-yaser aljohani
More from ProRelix Research
More from ProRelix Research (9)
Advancing Healthcare_ The Rise of Hybrid Virtual Clinical Trials.pdf

Advancing Healthcare_ The Rise of Hybrid Virtual Clinical Trials.pdf
Unlocking Opportunities in Online Clinical Research Courses.pdf

Unlocking Opportunities in Online Clinical Research Courses.pdf
Real-World Evidence Studies_ Introduction, Purpose, and Data Collection Strat...

Real-World Evidence Studies_ Introduction, Purpose, and Data Collection Strat...
Cancer Clinical Trials_ USA Scenario and Study Designs.pdf

Cancer Clinical Trials_ USA Scenario and Study Designs.pdf
Recently uploaded
Best medicine 100% Effective&Safe Mifepristion ௵+918133066128௹Abortion pills ...

Best medicine 100% Effective&Safe Mifepristion ௵+918133066128௹Abortion pills ...Abortion pills in Kuwait Cytotec pills in Kuwait
Recently uploaded (20)
Storage of Blood Components- equipments, effects of improper storage, transpo...

Storage of Blood Components- equipments, effects of improper storage, transpo...
Varicose Veins Treatment Aftercare Tips by Gokuldas Hospital

Varicose Veins Treatment Aftercare Tips by Gokuldas Hospital
Hemodialysis: Chapter 1, Physiological Principles of Hemodialysis - Dr.Gawad

Hemodialysis: Chapter 1, Physiological Principles of Hemodialysis - Dr.Gawad
Best medicine 100% Effective&Safe Mifepristion ௵+918133066128௹Abortion pills ...

Best medicine 100% Effective&Safe Mifepristion ௵+918133066128௹Abortion pills ...
NDCT Rules, 2019: An Overview | New Drugs and Clinical Trial Rules 2019

NDCT Rules, 2019: An Overview | New Drugs and Clinical Trial Rules 2019
SEMESTER-V CHILD HEALTH NURSING-UNIT-1-INTRODUCTION.pdf

SEMESTER-V CHILD HEALTH NURSING-UNIT-1-INTRODUCTION.pdf
Mgr university bsc nursing adult health previous question paper with answers

Mgr university bsc nursing adult health previous question paper with answers
Top 15 Sexiest Pakistani Pornstars with Images & Videos

Top 15 Sexiest Pakistani Pornstars with Images & Videos
VIII.1 Nursing Interventions to Promote Healthy Psychological responses, SELF...

VIII.1 Nursing Interventions to Promote Healthy Psychological responses, SELF...
Gross Anatomy and Histology of Tongue by Dr. Rabia Inam Gandapore.pptx

Gross Anatomy and Histology of Tongue by Dr. Rabia Inam Gandapore.pptx
parliaments-for-health-security_RecordOfAchievement.pdf

parliaments-for-health-security_RecordOfAchievement.pdf
Negative Pressure Wound Therapy in Diabetic Foot Ulcer.pptx

Negative Pressure Wound Therapy in Diabetic Foot Ulcer.pptx
CONGENITAL HYPERTROPHIC PYLORIC STENOSIS by Dr M.KARTHIK EMMANUEL

CONGENITAL HYPERTROPHIC PYLORIC STENOSIS by Dr M.KARTHIK EMMANUEL
High Purity 99% PMK Ethyl Glycidate Powder CAS 28578-16-7

High Purity 99% PMK Ethyl Glycidate Powder CAS 28578-16-7
Slider post_PR-07_14 Sep 1.pdf
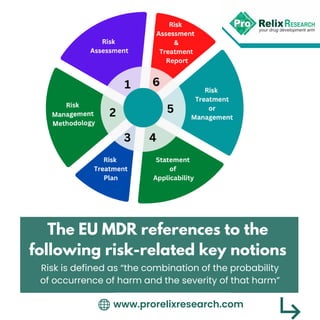
- 1. The EU MDR references to the following risk-related key notions www.prorelixresearch.com Risk is defined as “the combination of the probability of occurrence of harm and the severity of that harm”
- 2. www.prorelixresearch.com Benefit-Risk Determination is defined as “the analysis of all assessments of benefit and risk of possible relevance for the use of the device for the intended purpose, when used in accordance with the intended purpose given by the manufacturer”
- 3. General obligations are defined as “Manufacturers shall establish, document, implement and maintain a system for risk management” www.prorelixresearch.com
- 4. www.prorelixresearch.com QMS for Risk Management: The Quality Management Systems shall address the following matter – “risk management”
- 5. Establish and document a risk management plan for each device; Identify and analyze the known and foreseeable hazards associated with each device; Estimate and evaluate the risks associated with, and occurring during, the intended use and during reasonably foreseeable misuse; Eliminate or control the risks; The following requirements by the MDR should be addressed in order to ensure compliance and correct benefit/risk management: www.prorelixresearch.com
- 6. Evaluate the impact of information from the production phase and, in particular, from the post-market; Surveillance system, on hazards and the frequency of occurrence thereof, on estimates of their associated risks, as well as on the overall risk, benefit-risk ratio and risk acceptability; Amend control measures if necessary. www.prorelixresearch.com
- 7. ProRelix Research is here for the timely execution of your projects! www.prorelixresearch.com
