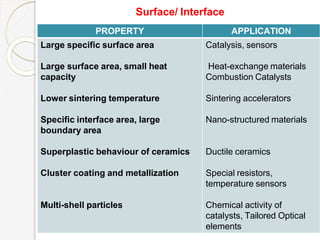
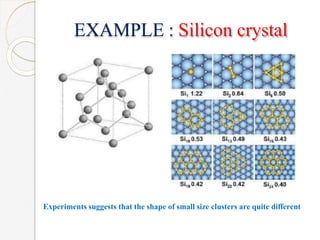
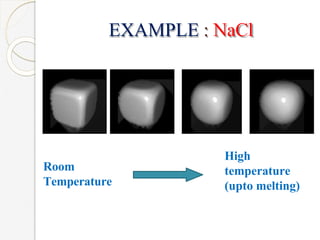
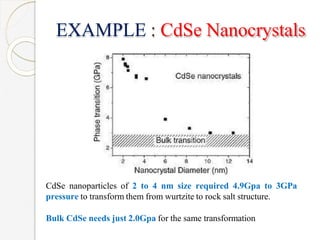
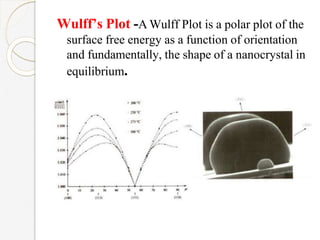
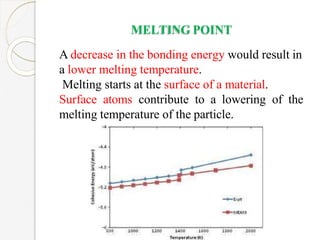
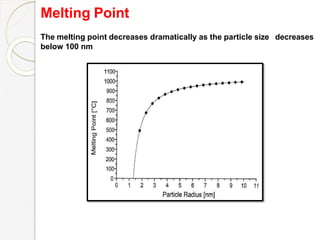
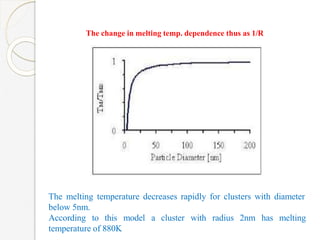
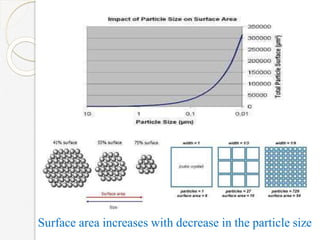
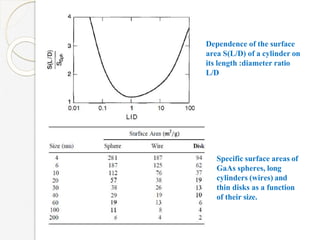
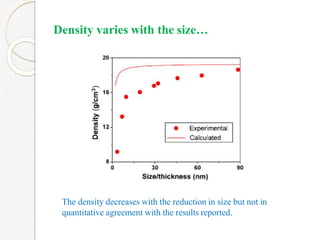
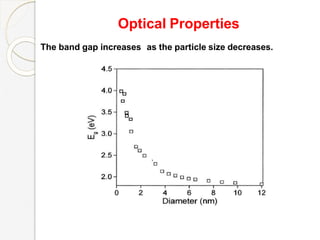
The document discusses the size effects of nanoparticles including their physical properties, shapes, and applications. It states that nanoparticles less than 100 nm exhibit size-dependent properties not seen in bulk materials, such as higher strength. The properties of nanoparticles can change with temperature and pressure due to changes in crystal structure. Their large surface area to volume ratio gives nanoparticles additional properties like improved catalytic activity.